As I commence my 2021 ANAT residency, it might be a good idea to introduce my project and provide some information regarding my background and the development of the project concept.
Apologies…this post is going to be a bit long…
As a visual artist I have always been really interested in the body in relation to technology including biotechnologies. During my PhD I was fortunate to have the opportunity to work across art and science at Queensland University of Technology and become actively integrated in the then Tissue Repair and Regeneration Group – a research team that focused on wound healing with expertise in cell and tissue culture. As part of this engagement, I was trained by team members and learnt how to culture different cells, human and animal, in a lab environment.
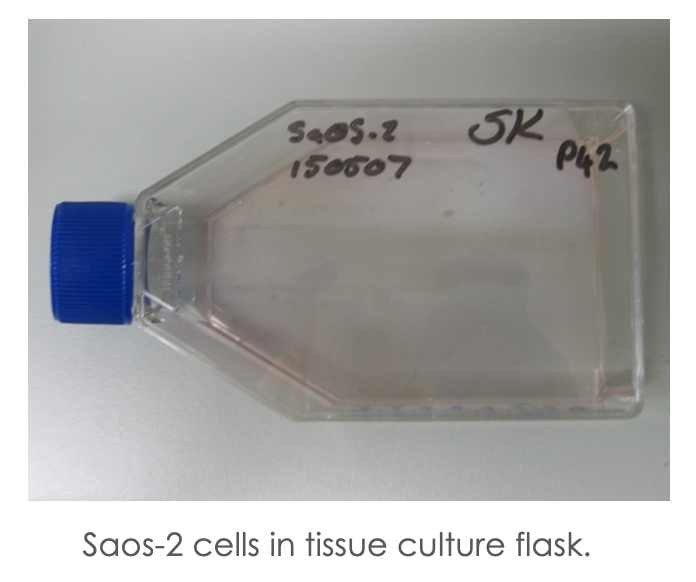 The first flask of cells that I received were Saos 2 cells – referencing their origin as sarcoma osteogenic – so a bone cancer cell line which was established in the 1970s from an 11-year old girl l, I named Alice.
The first flask of cells that I received were Saos 2 cells – referencing their origin as sarcoma osteogenic – so a bone cancer cell line which was established in the 1970s from an 11-year old girl l, I named Alice.
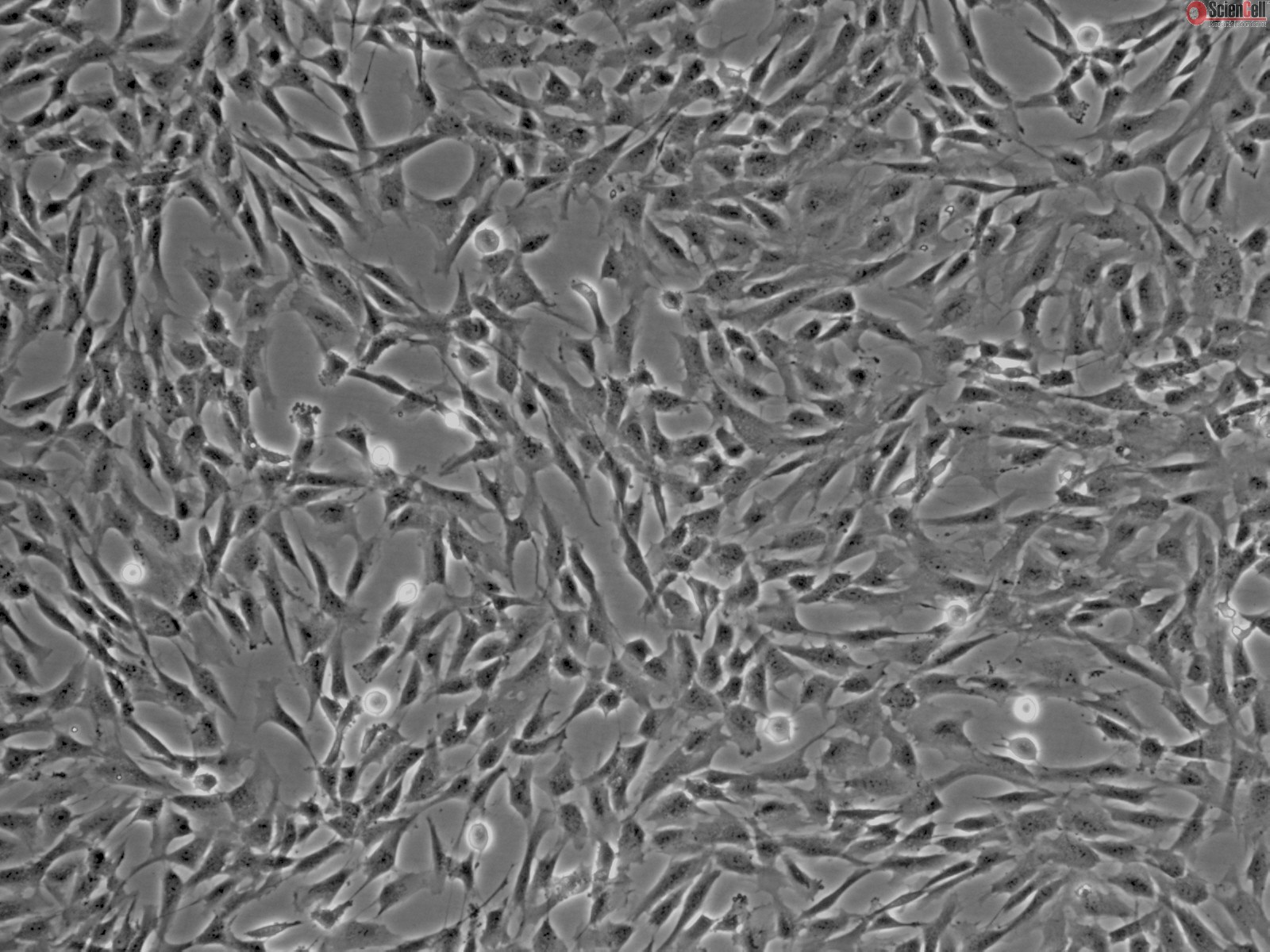
These cells, like many cell lines, are grown in a single layer on the base of tissue culture flasks with a liquid nutrient medium (see image above).
What struck me about these cells was that as cancer cells, they were essentially immortalised, in that they could reproduce well beyond the standard roughly 50 divisions reserved for healthy cells known as the Hayflick limit. As such, they can continue to replicate and be split into more and more tissue culture flasks as they fill up the available space – a process called passaging.
During this continued culture I observed that the cells are not inert but actually highly responsive to different culture conditions such as changes in media. Over the first 6 months or so, I split the original flask of cells 78 times and noted that the cells started to look quite different from the original cells I started with – they became sluggish and some of the cells even appeared to be multinucleated, so having more than one nucleus. That is also why lower passage cells – or cells that have not been split many times are preferred for research as they have less chance of mutation (genome and phenotype changes) and are more in line with the established characteristics of the cell line.
The first body of works that I developed in response to my experiences in the lab really reflected on the uncanniness of the Saos-2 cell line and the way in the cells are like living fragments of an absent body. The cells themselves also retain this intimate connection to the original donor as they contain Alice’s DNA, but at the same time they are representative of her disease and the longer they are cultured, the more they change and evolve. These observations were translated into the development of several interrelated works under the collective title The Absence of Alice that commented on the origins of the cell line but also foregrounded notions of continual becoming.
 My interest in cell lines was further developed during a residency at SymbioticA, which is a centre for excellence in the biological arts at UWA in 2000. As part of the engagement, I worked with HEK293T cells. These cells are also a cell line – so immortalised cells where the designation HEK refers to their origins as Human Embryonic Kidney cells. The number 293 documents the number of experiments required to immortalise the cells which was done using sheared adenovirus 5 DNA. Finally, the T signals a further alteration to the cells through the incorporation of the Simian virus 40 large T antigen referring to a specific protein from an animal virus. The addition of the protein rendered the cell line easier to transfect – so to introduce additional genetic material DNA or RNA into the cell.
My interest in cell lines was further developed during a residency at SymbioticA, which is a centre for excellence in the biological arts at UWA in 2000. As part of the engagement, I worked with HEK293T cells. These cells are also a cell line – so immortalised cells where the designation HEK refers to their origins as Human Embryonic Kidney cells. The number 293 documents the number of experiments required to immortalise the cells which was done using sheared adenovirus 5 DNA. Finally, the T signals a further alteration to the cells through the incorporation of the Simian virus 40 large T antigen referring to a specific protein from an animal virus. The addition of the protein rendered the cell line easier to transfect – so to introduce additional genetic material DNA or RNA into the cell.
What is interesting to note about the HEK cell line is that the original cell line HEK293 has since spawned a range of subcultures or daughter cells including 12 “tribes” catalogued by researchers in 2018 including HEK 293 T, of course, along with 11 others including HEK293S, HEK293F, HEK293FT and so on and there are even more now (Yuan, Jiang, Yu & Poon 2018).
What really struck me about working with the HEK cells was how an organism that never reached full gestation could nonetheless result in a whole series of unique microorganisms with different traits that as Oron Catts and Ionat Zurr from the Tissue Culture and Art Project (2006) assert start to resist easy “biological or cultural classifications”. Furthermore, the total biomass of the resulting cells often vastly outweighs the original body from which they were derived which is definitely the case for HEK cells and the various derivatives.
 The residency at Symbiotica also facilitated an introduction to basic genetic engineering processes and I learnt techniques to introduce red and green fluorescent proteins derived from marine invertebrates into the HEK cells which enabled them to glow red or green under specific light conditions.
The residency at Symbiotica also facilitated an introduction to basic genetic engineering processes and I learnt techniques to introduce red and green fluorescent proteins derived from marine invertebrates into the HEK cells which enabled them to glow red or green under specific light conditions.
These techniques are of course highly established processes used quite routinely in research labs for live cell imaging and cell marking, studying gene expression and observing the spread of disease and of course popularised in the art world by Eduardo Kac’s famous transgenic GFP Bunny project in which the artist claimed to have created the first glowing bunny in 2000.
Regardless, of the relatively routine use of fluorescent proteins in science and art, I found it fascinating to observe first-hand the ability to transfer genetic material from different species to facilitate new characteristics but also how these processes complicate traditional species and taxonomic distinctions – but also a recognition of how we – human and more-than-human – are kind of already set up to do this – to share genetic material.
One of the works produced as an outcome from the residency was the work HEK 293T: The transformation of Johni or Oliver which consisted of a mixed media sculpture and video work incorporating tissue culture flasks with fixed genetically engineered cells from the experiments conducted at Symbiotica. The work also included taxidermy butterflies and a range of unsettling elements merging the human and what would traditionally be regarded as non-human to highlight interconnections and potentials for further transformation, as well as the uncanny nature of bioengineered microorganisms that incorporate genetic material from viruses and other organisms and transgress traditional boundary distinctions.
 More recently, my work has increasingly focused on notions of immortality and the concept of genetic legacy. This interest was in many ways a move on from this earlier work with immortal cell lines but also the result of my own situation as a single woman in the later stages of reproductive viability. This prompted me to consider how I might be able to use new and emerging biotechnologies to establish an alternative genetic legacy beyond giving birth to human offspring.
More recently, my work has increasingly focused on notions of immortality and the concept of genetic legacy. This interest was in many ways a move on from this earlier work with immortal cell lines but also the result of my own situation as a single woman in the later stages of reproductive viability. This prompted me to consider how I might be able to use new and emerging biotechnologies to establish an alternative genetic legacy beyond giving birth to human offspring.
The initial exploration was inspired by a range of emerging medical processes such as genetic engineering and gene therapy which allow the integration of genetic material into host organisms alongside scientific insights in relation to the presence of viral DNA in the human genome. For example, it now largely accepted that humans contain up to 8% viral DNA from ancestral infection and that the integration of viral information may have facilitated the transition to placental birth in mammals – illustrating that viruses likely have a significant role to play in the evolution of organisms.
Drawing on these ideas, I developed the work Self-Portrait #2: Site of Infection for the 2019 show Pandemic curated by Toby Gifford at the Plimsoll Gallery in Hobart – developed to coincide with the 100 -year anniversary of the 1918–1919 global Spanish influenza pandemic but in retrospect the show was also a strange predictor for the 2019 – 2020 outbreak of COVID-19.

The work I produced consisted of a wax cast of my bust rendered in a style reminiscent of classical depictions of female deities that emits vapour containing my DNA. By incorporating this DNA breath, the work alludes to the idea of using viral infection as a potential mechanism of proliferating my genetic material. Essentially, I was speculating that by genetically engineering viruses to include fragments from my genetic code, it may be possible to infect other organisms including humans and use the virus as a mechanism to reproduce a bit of my genetic material within the selected host’s body. I also thought that by using a retrovirus and targeting germ cells such as sperm, eggs, it might be possible that the selected DNA sequences could be passed on to the organism’s offspring.
In this way, I could pass on a portion of my genetic information without having children of my own – because other people and their children would be hosts and continue to perpetuate a piece of me in perpetuity.
The work was designed to be deliberately provocative and highlight the ‘uncomfortability’ many of us experience at the prospect of infection (especially now) and the idea of ‘genetic contamination’ despite my view which follows Timothy Moreton’s (2010) assertion that we are not so much individuals but rather part of a mesh or multi-species ecologies comprised of shared and swapped genetic material.
More recently, my work has returned to cell culture and particularly the prospect of establishing an immortalised cell line from my own body via a range of processes such as using viral genes such as using SV40 T antigens to disrupt programmed senescence and cell death.
While, there is a processor, the Billy Apple ® cell line devised by artist Craig Hilton (2014) in 2010, the desire to produce my own cell line developed from my own recent experience of uterine pathology.
Essentially, the proposition follows a two-year stint of being very, very unwell. After quite a long delay, In 2019, I was diagnosed with a large fibroid, which is essentially a benign tumorous growth of the uterus that is quite common in women of later reproductive age.
![Tumour Baby [Fibroid]](https://kratz2021.blog.anat.org.au/files/2021/06/tumourbaby.jpg)
To relieve a range of symptoms, I needed to undergo surgery to remove the mass and saw this as an opportunity to not only harvest tissue from the fibroid, but also use the project as a mechanism to explore the prevailing cultural attitudes towards women in relation to fertility and motherhood and use my experiences of seeking a diagnosis to reveal how women are often treated in a medical domain.
For example, despite having a really great GP it still took a while to receive a diagnosis because period pain, even extreme pain, is often dismissed, as something so common it generally does not really warrant further investigation.

It was only when I had accumulated evidence such as not realising that I broke my arm because the pain was insignificant in comparison to period pain and collecting huge clots of blood that it became apparent that my situation warranted a formal scan.
When I was diagnosed, the initial assumption by medical staff seemed to be that preserving fertility was not really a necessity because of my age evidenced with some of the initial treatments suggested such as ablation – which though less invasive would have likely compromised my ability to conceive, but also hysterectomy to relieve the symptoms. It also made me very aware that despite the relatively low chance of having children, it was an expected future that I was not ready yet to give up on all together.
Prior to having the tumour removed, I developed a series of preliminary works for a solo show titled Mourning Story: Expectations, Absences and the Potentials for Self-Persistence that attempted to give some insight into my experiences of situational childlessness and the grief that can accompany the experience but also the problematic constructions of womanhood that reinforce traditional gender expectations including assumed motherhood.

One of the first works developed as part of the series consisted of four marquetry panels developed with Anita Gowers and Phil Blacklow. The panel designs referenced floral motives centred on the Victorian language of flowers dictionary in relation to femininity. The panel featured on this slide for example references Ivy – which denotes marriage and fidelity. The centre of the panel features the clotted period blood collected pre-diagnosis.

After Spring also developed in collaboration with Anita and Phil follows this line of engagement and references work by the Renaissance painter Sandro Botticelli which I feel effectively captures Western mythological constructions of an idealised femininity.

The painting references Primavera, however the central figure is from The Calumny of Apelles representing truth. She points upwards at 3D model of a scallop referencing Botticelli’s The Birth of Venus. In this instance, the scallop is not a neat and clean construction, but shows the veins, digestive system making a comment on notions of ideal and myth vs. wet bodily reality. The central figure was also rendered as an anatomical Venus referencing the way in which medical imagery and knowledge is influenced by cultural representations. The panels either side of the painting show ripples representing the way in which these cultural constructions continue to ripple through time and influence contemporary perceptions and knowledge.
The work was exhibited in conjunction with Time is Topological, which included a series of ticking clocks and a repurposed bleeding table displaying various items including an anatomical model with a snake in its belly. This element references the legacy of hysteria and wandering womb – the belief that the uterus wanders about body of women like a wild animal and it is this condition that causes major illnesses including hysteria in women – a view documented in medical texts from ancient Greece including the Hippocratic Corpus, and as Amy Koerber (2018) points out in her book From Hysteria to Hormones has seen various manifestations and evolved to the 20th century view where women’s so-called irrationality is linked to hormones.

Other works such as the Memento Mori series which consisted of a forest of dome works representing death, transformation and hope and the life-size sculptural work Coming to Terms with Being Forgotten. These works give insight into feelings of mourning for lost possibilities, but also a recognition that because we are already connected into a long history of genetic continuance that effectively links us to all other organisms and deep time histories, that the idea of establishing a genetic legacy is really unnecessary and that maybe our role is really to die and form part of a larger cycle that extends beyond ourselves.

The final work in the series, SVKR-LM: Tumour Baby returns back to my original desire to establish a cell line and alludes to the current stage of project development which aims to establish a cell line from the fibroid tissue. By incorporating a flask of fixed primary skin cells of mine, established from another project, and incorporating taxidermy butterflies and uncanny elements, the work makes a direct reference back to the work HEK293T: The Transformation of Johni or Oliver signalling the potential for the fibroid cells to not only establish a single cell line but spawn a range or tribe of daughter cells that like the intention of the Billy Apple ® cell line could be used for both scientific and creative research endeavours.

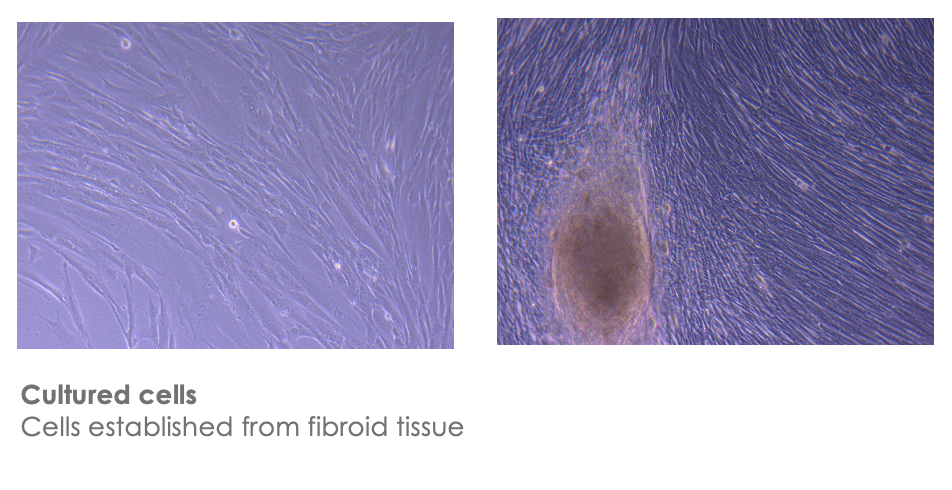
Since this body of work was shown, my fibroid has been removed and I was fortunate to receive ethics clearance in time and find collaborators from the school of medicine A/prof Brad Sutherland and Dr Jo-Maree Courtney who have isolated, cultured and frozen down cells from the tissue.
Here you can see images of the isolated cells which look like fibroblasts to me, but also because fibroblasts are really common in skin and connective tissue in animals.
 That brings us up to date in terms of the status of the project. The ANAT Synapse grant will enable me to work closely with Brad and Jo-Maree from the School of Medicine, alongside A/Prof Jane Nielsen from Centre for Law and Genetics at UTAS and Prof Dietmar Hutmacher from the Regenerative Medicine Group at QUT on hopefully establishing the second artist cell line for inclusion in the American Type Culture Collection or ATCC (the central global repository for cell lines). I am really interested in using the process of establishing the cell line to examine current legal and ethical frameworks for the management of use of biological materials across multiple contexts (creative, clinical and commercial) including patent and IP considerations. Moreover, the project will also provide an opportunity to rethink the boundaries of the body and self and conceptualize alternative notions of kinship and care and how this might operate in a lab space that could be regarded as again my colleagues Oron and Ionat have observed as exploitative of the ‘semi-living’.
That brings us up to date in terms of the status of the project. The ANAT Synapse grant will enable me to work closely with Brad and Jo-Maree from the School of Medicine, alongside A/Prof Jane Nielsen from Centre for Law and Genetics at UTAS and Prof Dietmar Hutmacher from the Regenerative Medicine Group at QUT on hopefully establishing the second artist cell line for inclusion in the American Type Culture Collection or ATCC (the central global repository for cell lines). I am really interested in using the process of establishing the cell line to examine current legal and ethical frameworks for the management of use of biological materials across multiple contexts (creative, clinical and commercial) including patent and IP considerations. Moreover, the project will also provide an opportunity to rethink the boundaries of the body and self and conceptualize alternative notions of kinship and care and how this might operate in a lab space that could be regarded as again my colleagues Oron and Ionat have observed as exploitative of the ‘semi-living’.
Catts, O & Zurr, I 2006. Towards a new class of being: the extended body. Artnodes, 6(2), pp.1-9.
Hilton, C. 2014. The immortalisation of Billy Apple®: an art-science collaboration. Leonardo, 47(2), pp.109-113.
Koerber, A., 2018. From Hysteria to Hormones: A Rhetorical History , Penn State Press.
Morton, T 2010. The Ecological Thought. Harvard University Press.
Yuan J, Xu WW, Jiang S, Yu H, Poon HF 2018 The scattered twelve tribes of HEK293. Biomedical and Pharmacology Journal. 2018 June 25;11(2):621-3.



 Now that I am officially part of the Stroke Group (with Synapse collaborators A/Prof Brad Sutherland and Dr Jo-Maree Courtney), I attended my first Tuesday afternoon meeting. Brad was the presenting speaker who shared some of his research from a Postdoc undertaken at the University of Oxford some years ago. The talk focused on “Glutathione and its response following stroke”. While some of the discussion (particularly in relation to the Glutathione pathway) was beyond my knowledge, it was still useful to consider the discussion in terms of experimental design. It was also great to be reminded that sometimes outcomes are unexpected or unsatisfying, as they may contradict expectations.
Now that I am officially part of the Stroke Group (with Synapse collaborators A/Prof Brad Sutherland and Dr Jo-Maree Courtney), I attended my first Tuesday afternoon meeting. Brad was the presenting speaker who shared some of his research from a Postdoc undertaken at the University of Oxford some years ago. The talk focused on “Glutathione and its response following stroke”. While some of the discussion (particularly in relation to the Glutathione pathway) was beyond my knowledge, it was still useful to consider the discussion in terms of experimental design. It was also great to be reminded that sometimes outcomes are unexpected or unsatisfying, as they may contradict expectations.
 The first flask of cells that I received were Saos 2 cells – referencing their origin as sarcoma osteogenic – so a bone cancer cell line which was established in the 1970s from an 11-year old girl l, I named Alice.
The first flask of cells that I received were Saos 2 cells – referencing their origin as sarcoma osteogenic – so a bone cancer cell line which was established in the 1970s from an 11-year old girl l, I named Alice. My interest in cell lines was further developed during a residency at
My interest in cell lines was further developed during a residency at  The residency at Symbiotica also facilitated an introduction to basic genetic engineering processes and I learnt techniques to introduce red and green fluorescent proteins derived from marine invertebrates into the HEK cells which enabled them to glow red or green under specific light conditions.
The residency at Symbiotica also facilitated an introduction to basic genetic engineering processes and I learnt techniques to introduce red and green fluorescent proteins derived from marine invertebrates into the HEK cells which enabled them to glow red or green under specific light conditions. More recently, my work has increasingly focused on notions of immortality and the concept of genetic legacy. This interest was in many ways a move on from this earlier work with immortal cell lines but also the result of my own situation as a single woman in the later stages of reproductive viability. This prompted me to consider how I might be able to use new and emerging biotechnologies to establish an alternative genetic legacy beyond giving birth to human offspring.
More recently, my work has increasingly focused on notions of immortality and the concept of genetic legacy. This interest was in many ways a move on from this earlier work with immortal cell lines but also the result of my own situation as a single woman in the later stages of reproductive viability. This prompted me to consider how I might be able to use new and emerging biotechnologies to establish an alternative genetic legacy beyond giving birth to human offspring.

![Tumour Baby [Fibroid]](https://kratz2021.blog.anat.org.au/files/2021/06/tumourbaby.jpg)







 That brings us up to date in terms of the status of the project. The ANAT Synapse grant will enable me to work closely with Brad and Jo-Maree from the School of Medicine, alongside A/Prof Jane Nielsen from Centre for Law and Genetics at UTAS and Prof Dietmar Hutmacher from the Regenerative Medicine Group at QUT on hopefully establishing the second artist cell line for inclusion in the American Type Culture Collection or ATCC (the central global repository for cell lines). I am really interested in using the process of establishing the cell line to examine current legal and ethical frameworks for the management of use of biological materials across multiple contexts (creative, clinical and commercial) including patent and IP considerations. Moreover, the project will also provide an opportunity to rethink the boundaries of the body and self and conceptualize alternative notions of kinship and care and how this might operate in a lab space that could be regarded as again my colleagues Oron and Ionat have observed as exploitative of the ‘semi-living’.
That brings us up to date in terms of the status of the project. The ANAT Synapse grant will enable me to work closely with Brad and Jo-Maree from the School of Medicine, alongside A/Prof Jane Nielsen from Centre for Law and Genetics at UTAS and Prof Dietmar Hutmacher from the Regenerative Medicine Group at QUT on hopefully establishing the second artist cell line for inclusion in the American Type Culture Collection or ATCC (the central global repository for cell lines). I am really interested in using the process of establishing the cell line to examine current legal and ethical frameworks for the management of use of biological materials across multiple contexts (creative, clinical and commercial) including patent and IP considerations. Moreover, the project will also provide an opportunity to rethink the boundaries of the body and self and conceptualize alternative notions of kinship and care and how this might operate in a lab space that could be regarded as again my colleagues Oron and Ionat have observed as exploitative of the ‘semi-living’.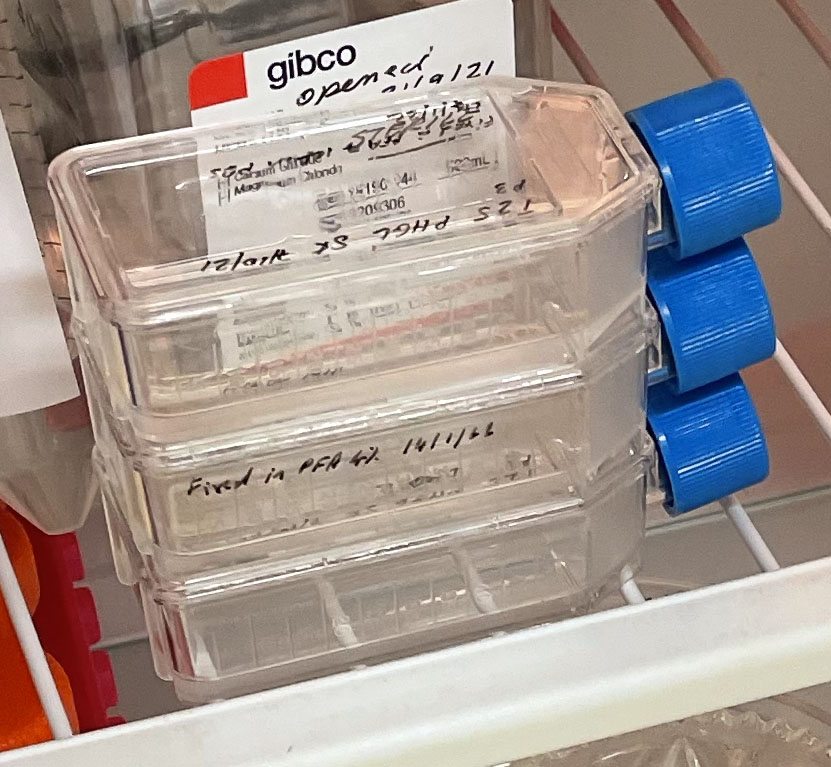 Three T25 PHGL Tumour Baby Flasks fixed with 4% PFA in PBS ready for staining.
Three T25 PHGL Tumour Baby Flasks fixed with 4% PFA in PBS ready for staining.