The lab has a great set-up for fluorescence microscopy which makes imaging quick and easy.
You just need to load the well plate into the machine and set up basic imaging parameters. You do need to image both DAPI and Phalloidin stains, but the software merges the images for you.
Fluorescent Images of Fibroid Cells
 Simple graphic interface with presets ready to complete fluorescent microscopy.
Simple graphic interface with presets ready to complete fluorescent microscopy.
As discussed in my previous post on immunostaining, the blue dots indicate nuclei and the green structures reveal the cytoskeleton via binding to actin.
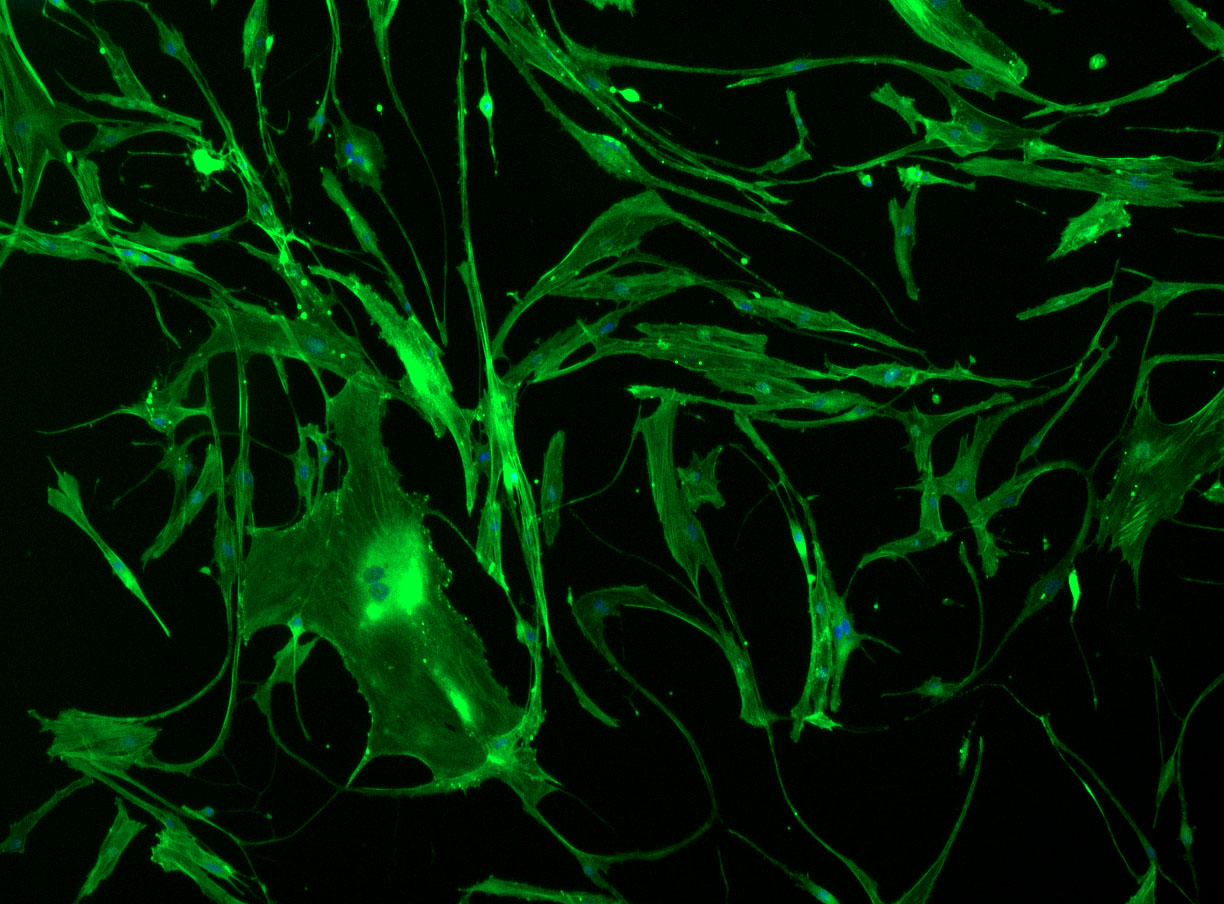
Confluent wells:
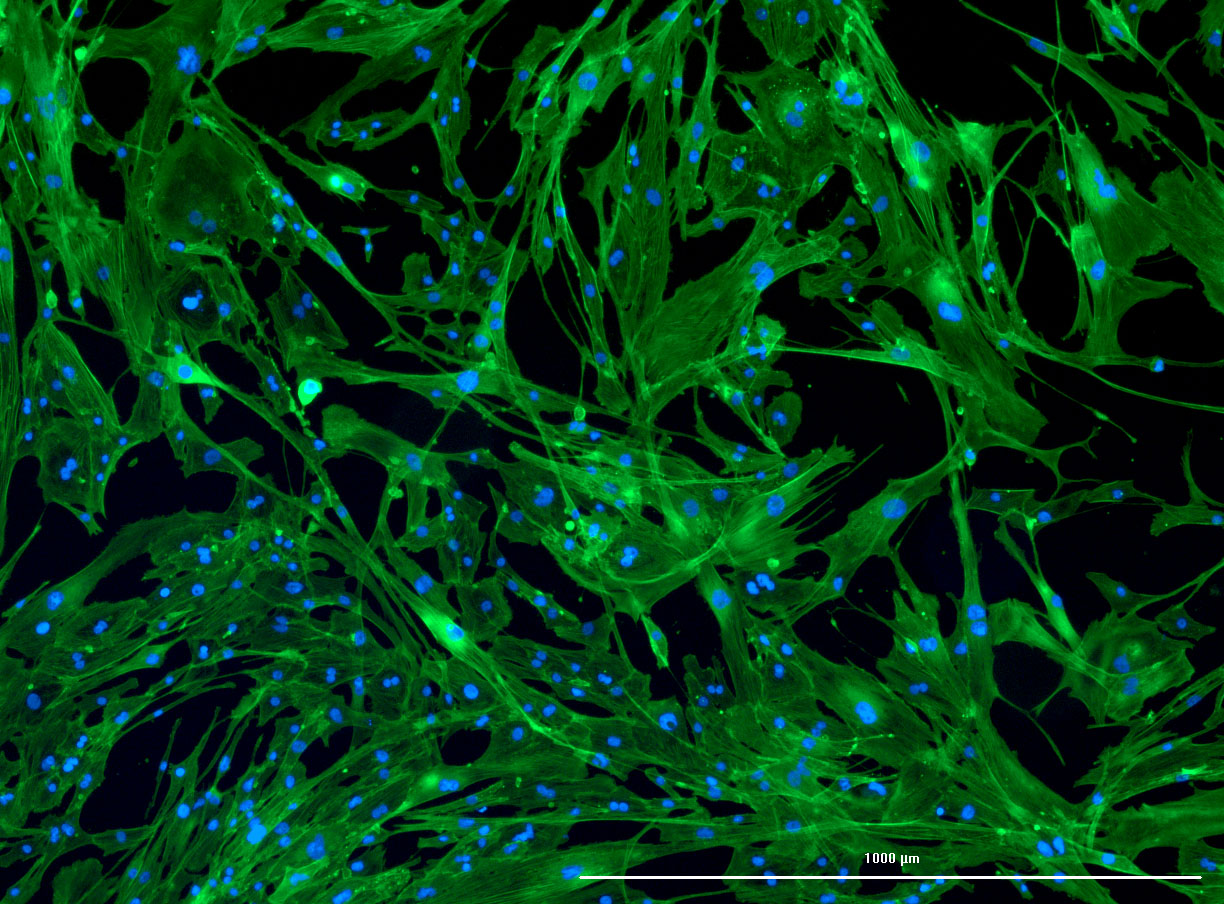
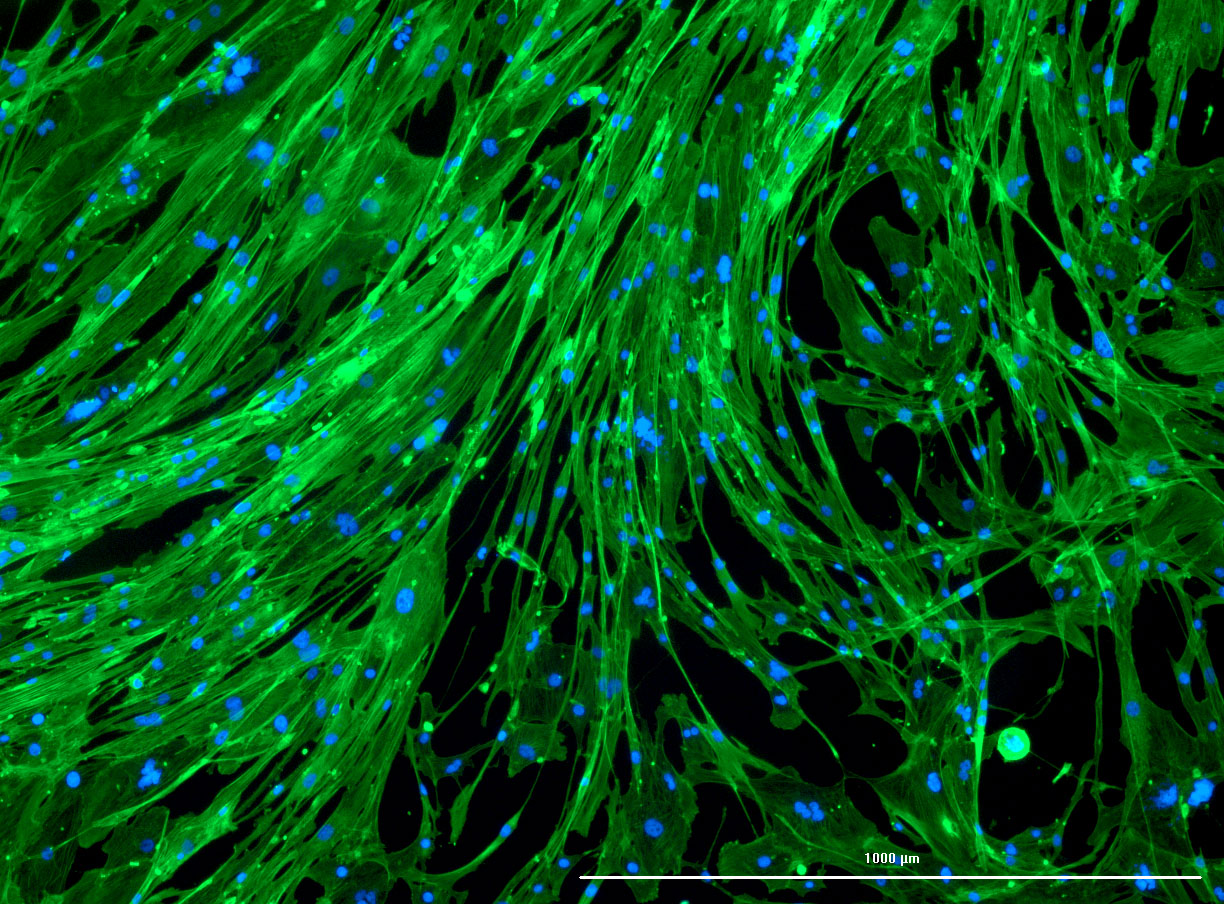
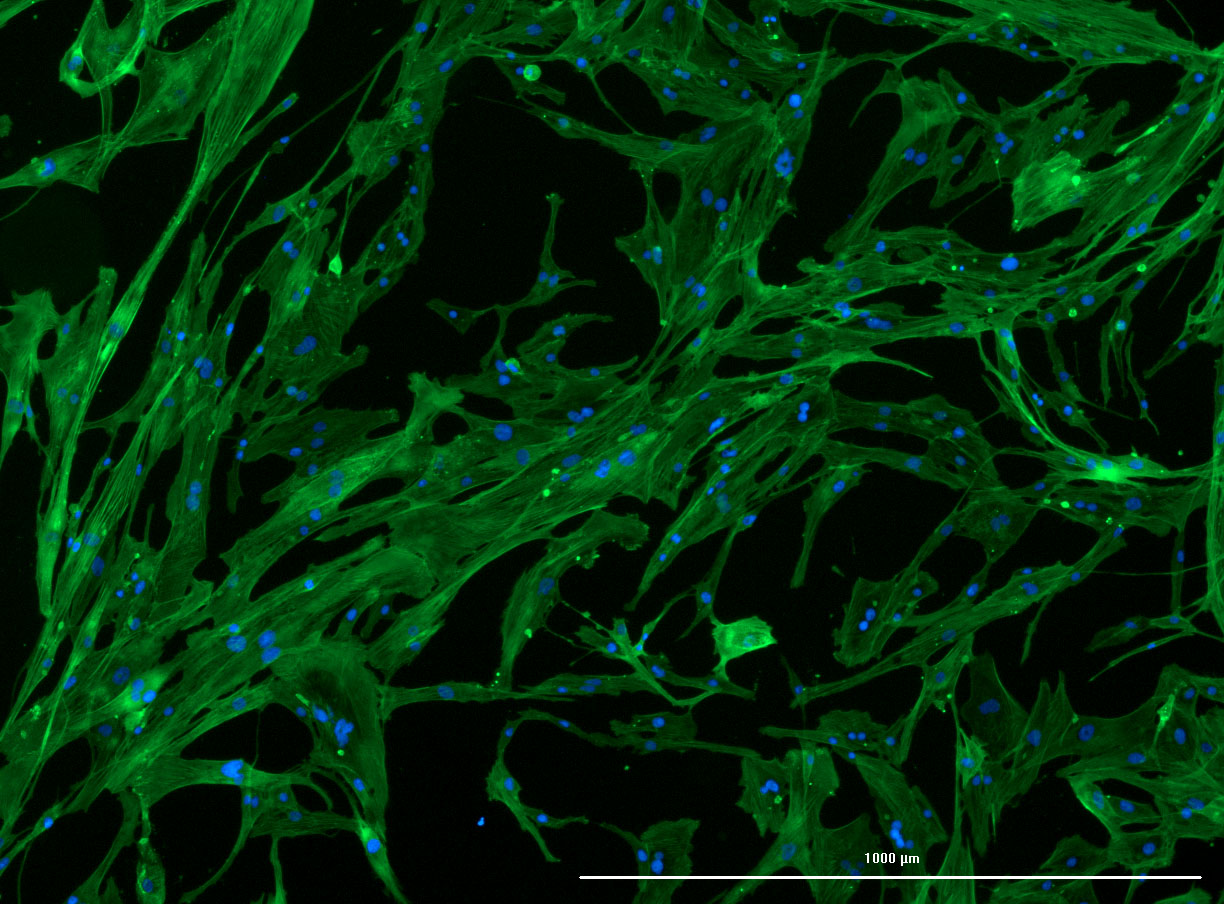 DAPI and Fluorescein Phalloidin staining of confluent fibroid cells P4 (although this is potentially misleading as the cells are very slow growing).
DAPI and Fluorescein Phalloidin staining of confluent fibroid cells P4 (although this is potentially misleading as the cells are very slow growing).
Less confluent wells:
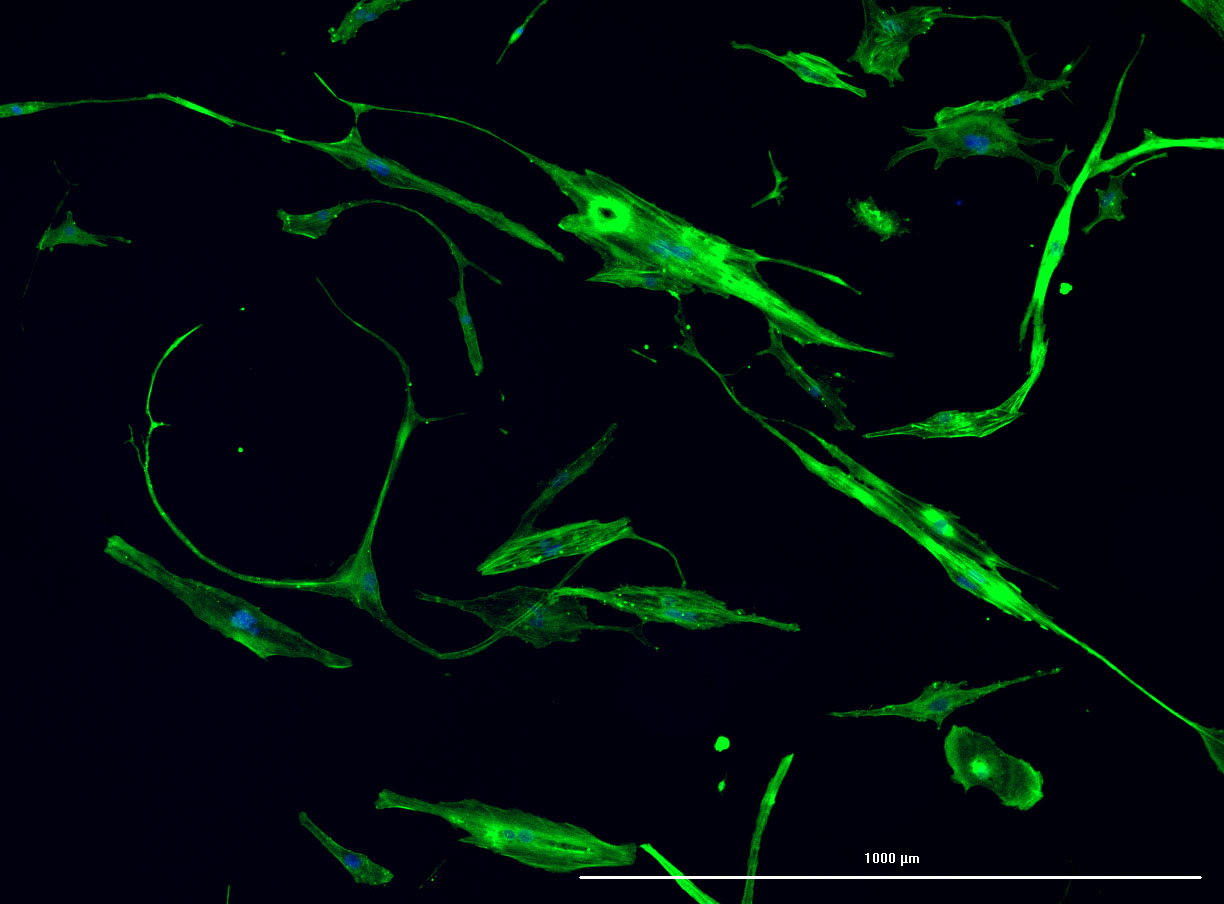
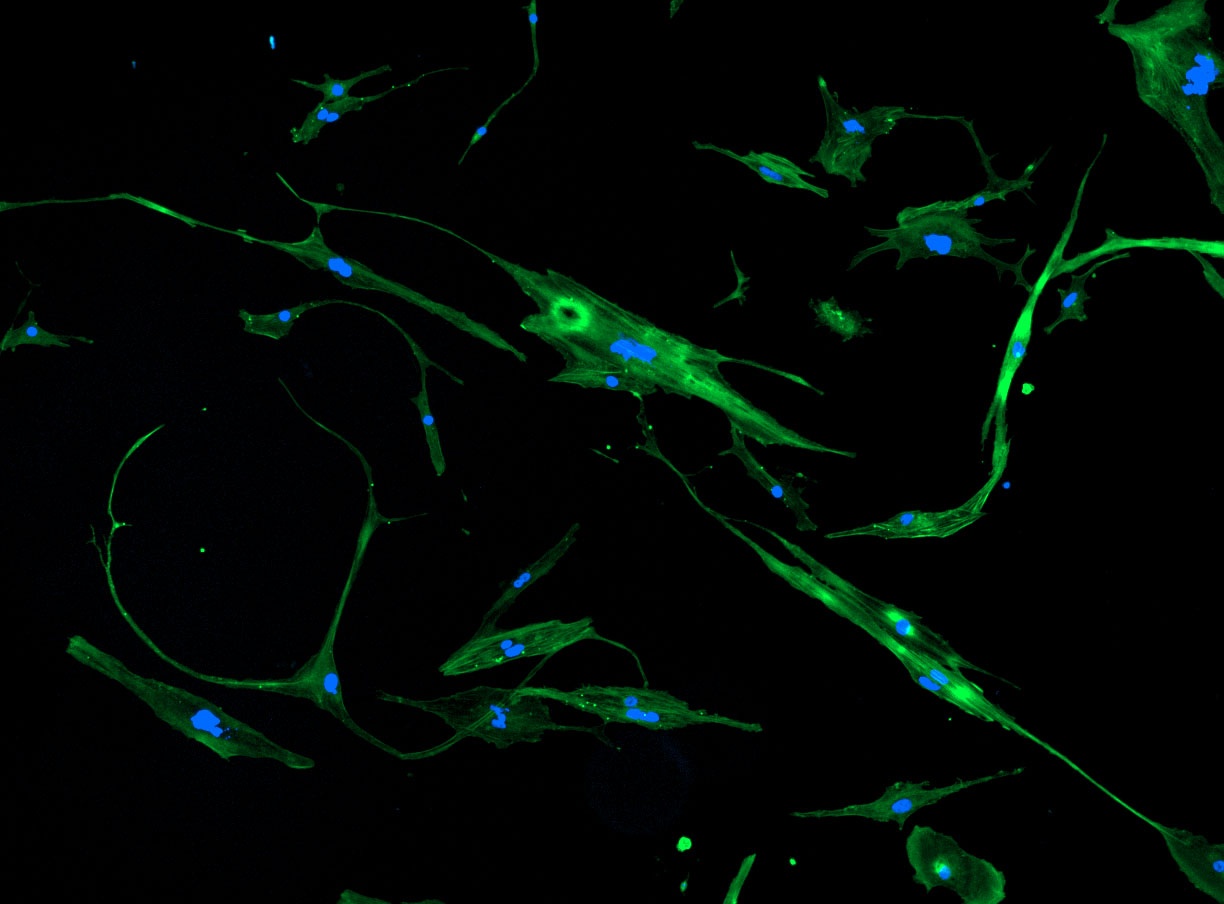
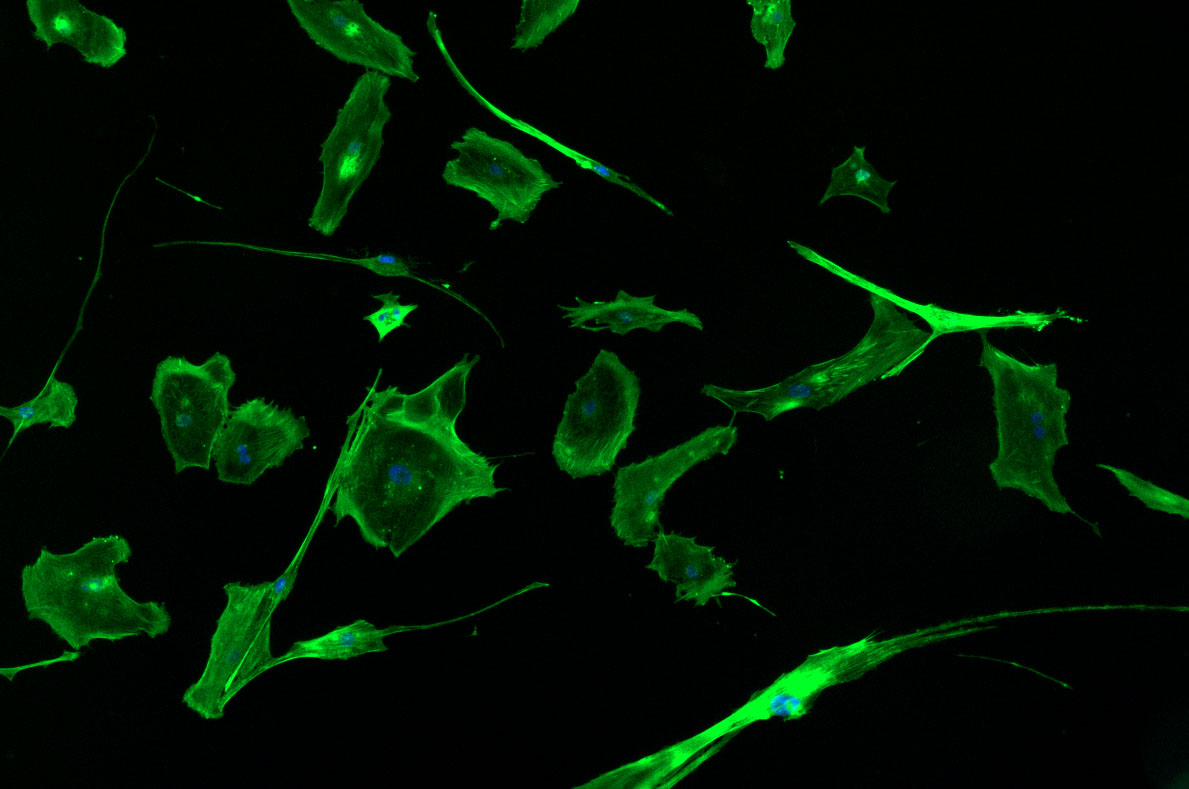
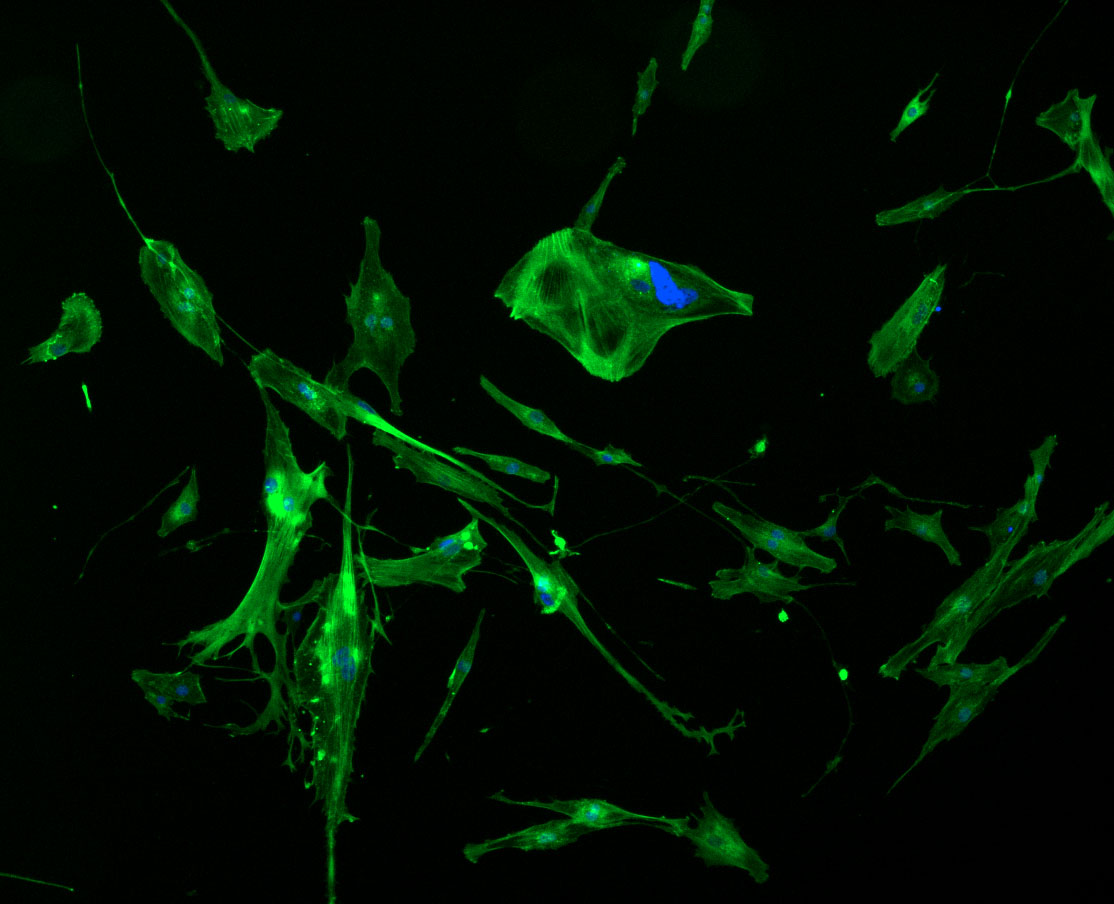
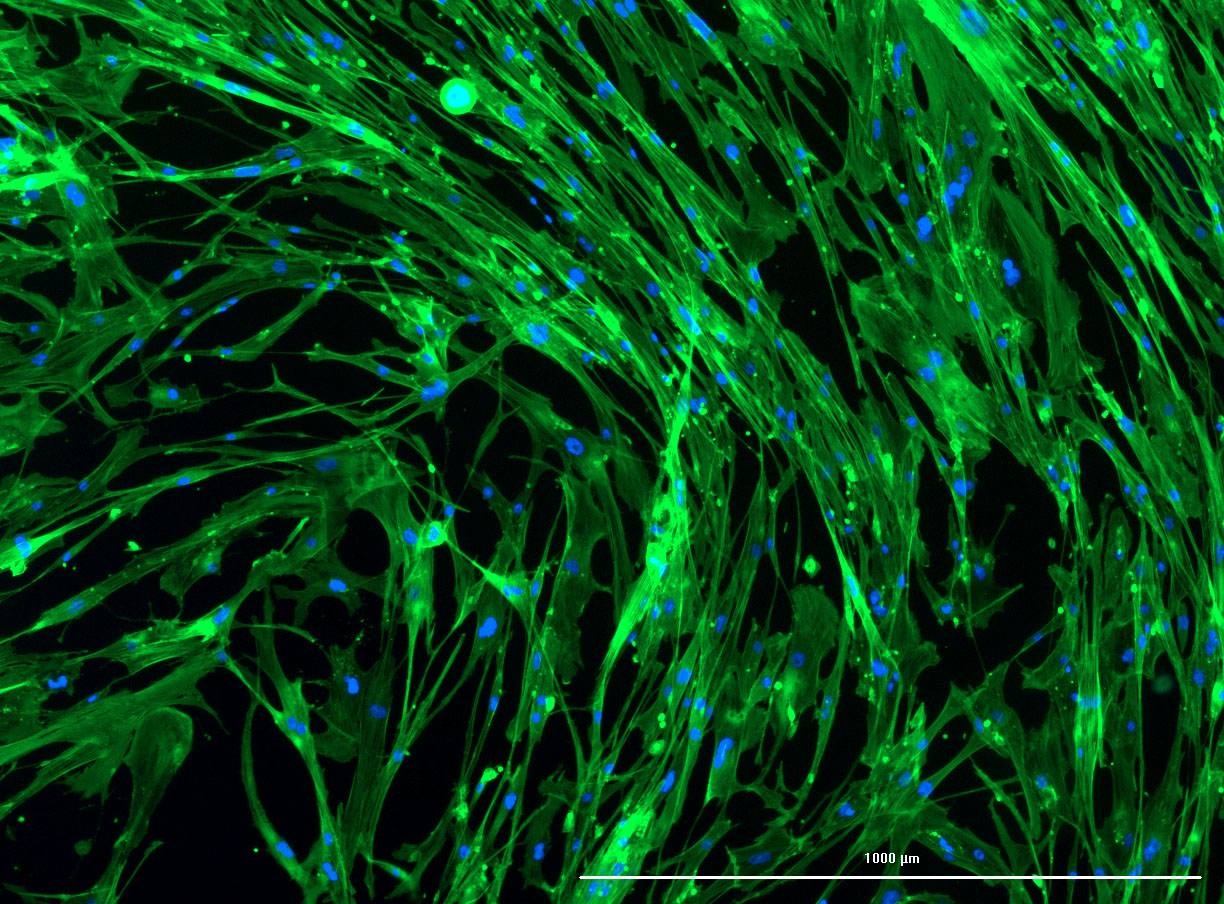
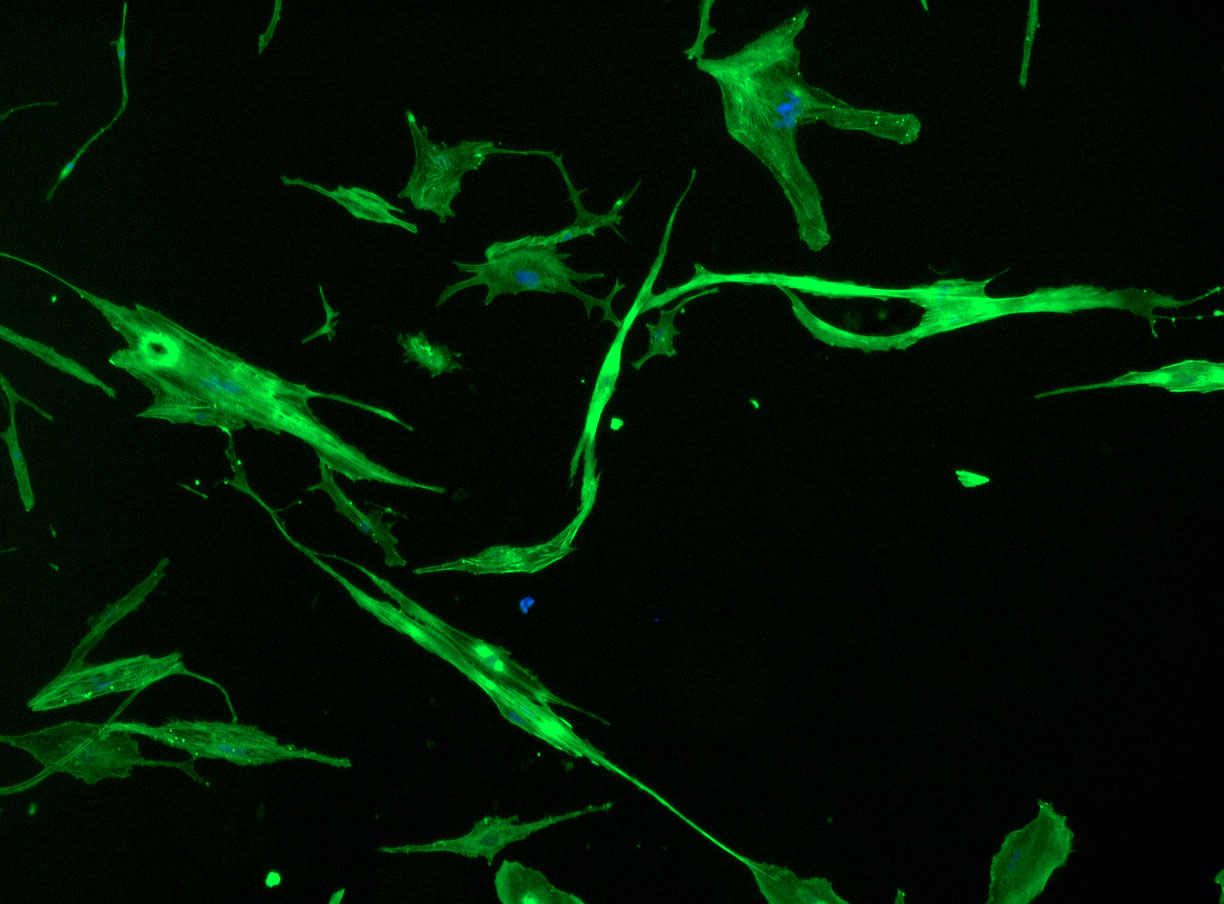 DAPI and Fluorescein Phalloidin staining of fibroid cells P4 which enables better visualisation of individual cells.
DAPI and Fluorescein Phalloidin staining of fibroid cells P4 which enables better visualisation of individual cells.
It is time to complete the immunostaining protocol with guidance from Jo-Maree. I must admit that with holidays looming, my note taking was a bit sketchy. I will need to follow up with Jo-Maree to record the correct details of DAPI and Fluorescein Phalloidin stain. This will ensure that I know how to prepare (and order) stocks in the future.
I already prepared a couple of wells at different cell concentrations ready for staining. We had to delay the protocol, so some of the higher concentration wells are likely a bit over-confluent. It will be interesting to see how they look under the fluorescence microscope.
CELL FIXATION: ‘Dirty’ Biolab
 Working with 4% PFA in Fume Hood
Working with 4% PFA in Fume Hood
Prior to imaging, I fixed the cells in 4% PFA:
- Remove culture media (discard in waste container with bleach)
- Wash cells with PBS (discard in waste container with bleach) x 2
- Move cells to fume hood
- Add 4% PFA to each well for 15 – 20min at room temp (in fume hood)
- Remove 4% PFA solution (discard in PFA waste container in fume hood)
- Add PBS (make sure cells are covered or they dry out and produce poor images
IMMUNOSTAINING: At lab bench area
 Working at lab bench in the Stroke Group area
Working at lab bench in the Stroke Group area
- Remove PBS from each well
- Add 1mL 0.3% Triton X-100 (a strong detergent) to permeabilize cells (make cells permeable – this allows the phalloidin stain to enter the cell structure) for 10 min
- Make up DAPI (5mL PBS Tween and 1ul DAPI) and protect from light with aluminium cover
- Remove Triton X-100 and add 1mL DAPI solution to each well and incubate at room temp (with aluminum cover to protect from light) for 5 minutes

- Make up Flouroscein Phalloidin (1mL PBS sand 2μL Flouroscein) and protect from light
- Remove and discard DAPI solution
- Add 1mL PBS 0.1% Tween to each well for 5 min then discard x 3 (i.e. wash with PBS Tween x 3)
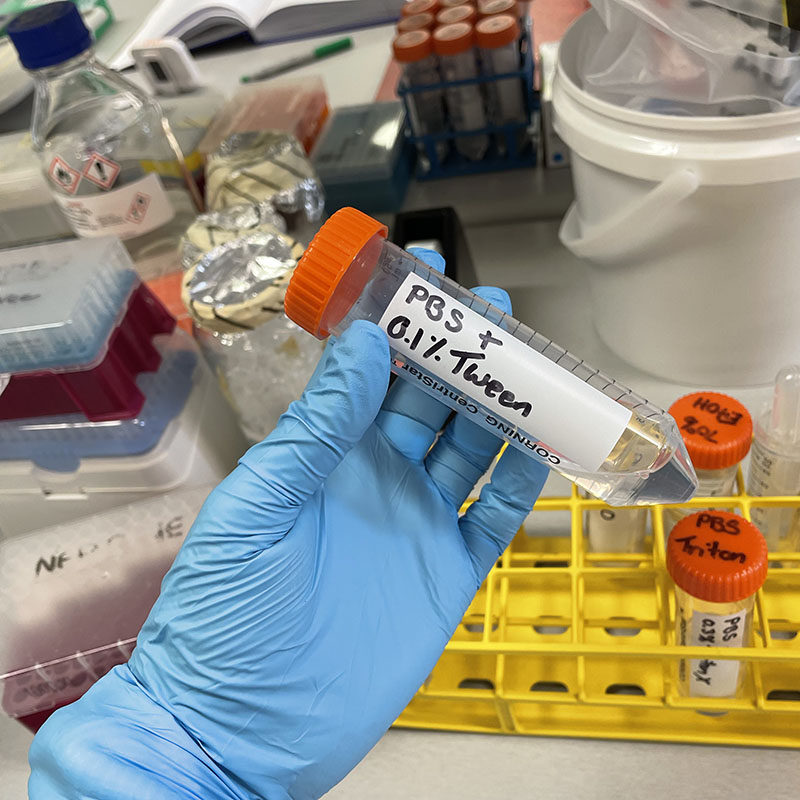
- Add Phalloidin stain to each well.
- Incubate at room temp for 1 – 2 hours
- Remove Phalloidin solution
- Wash with PBS Tween x 2
- Add 1mL PBS in each well
- Cover with aluminum foil to protect from light.
- Cells are ready for imaging. They can be stored in the fridge (with aluminium foil cover) until ready.
I think I can officially label myself a hoarder of materials – I never want to let anything go in case it may be creatively useful or yield something. As such, I have been maintaining most of my passage flasks hoping for cell survivors (and, if I’m honest, spontaneous mutation and cell immortalisation).
Today the time has come to let go of some of my flasks. I am building up to the task, so will start with some of my smaller flasks – the T25s.
Following the standard fixation protocol in 4% PFA, I’ve prepared them for staining with H&E. It will be interesting to see if there were any remaining cells and if the ‘ghost trails’ stain with Eosin.
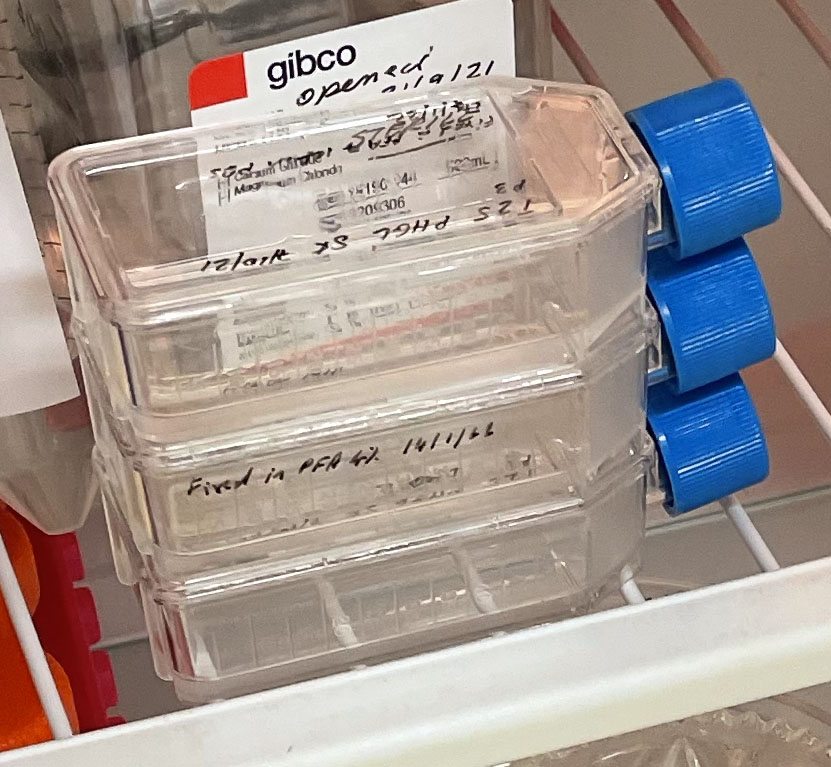 Three T25 PHGL Tumour Baby Flasks fixed with 4% PFA in PBS ready for staining.
Three T25 PHGL Tumour Baby Flasks fixed with 4% PFA in PBS ready for staining.
Recipient of ANAT Synapse Residency 2021

 Simple graphic interface with presets ready to complete fluorescent microscopy.
Simple graphic interface with presets ready to complete fluorescent microscopy. 


 DAPI and Fluorescein Phalloidin staining of confluent fibroid cells P4 (although this is potentially misleading as the cells are very slow growing).
DAPI and Fluorescein Phalloidin staining of confluent fibroid cells P4 (although this is potentially misleading as the cells are very slow growing). 




 DAPI and Fluorescein Phalloidin staining of fibroid cells P4 which enables better visualisation of individual cells.
DAPI and Fluorescein Phalloidin staining of fibroid cells P4 which enables better visualisation of individual cells. 
 Working with 4% PFA in Fume Hood
Working with 4% PFA in Fume Hood Working at lab bench in the Stroke Group area
Working at lab bench in the Stroke Group area

 Three T25 PHGL Tumour Baby Flasks fixed with 4% PFA in PBS ready for staining.
Three T25 PHGL Tumour Baby Flasks fixed with 4% PFA in PBS ready for staining.