My Tumour Baby cells still remain very sluggish and slow to replicate. I’ve been checking in regularly to chart their growth.
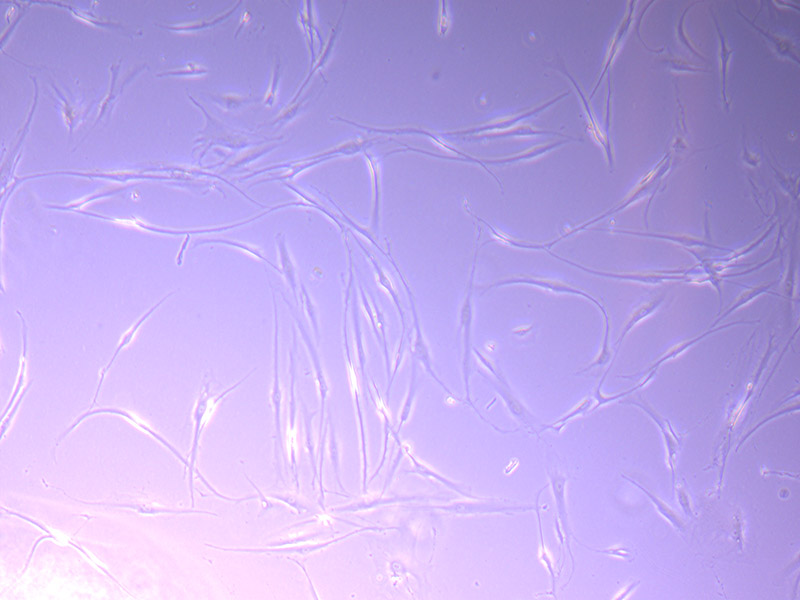
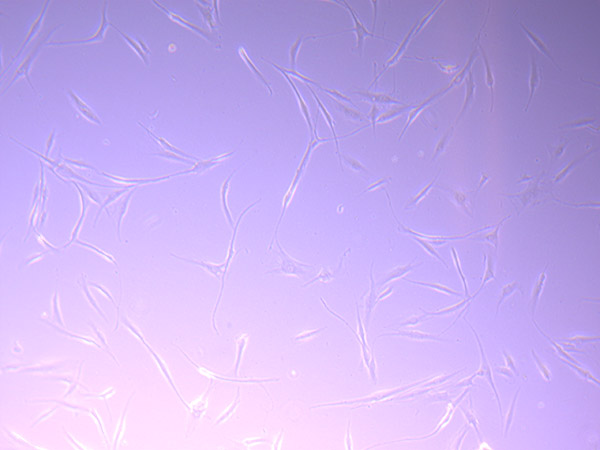
Brightfield microscope images of PHGL TB Cell Growth 17/09/21
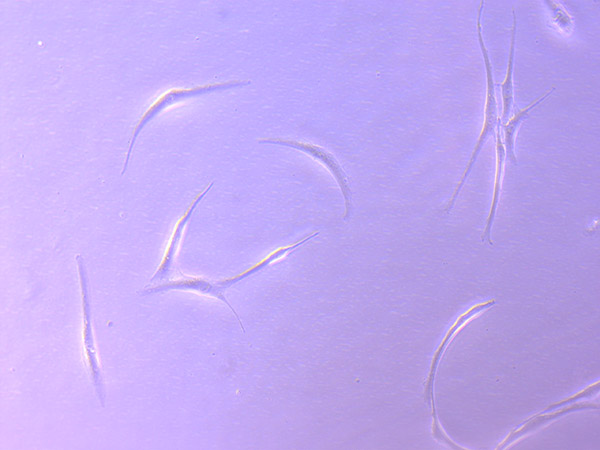
As they have continued to grow, they have started to look less healthy and consistent. They remind me of gamma irradiated 3T3 cells (mouse fibroblasts that have been irradiated to stop replicating). However, this could also be the result of using media that is not ideal, as we have been using some existing (expired) stock supplies of DMEM while we are waiting for our order to arrive.
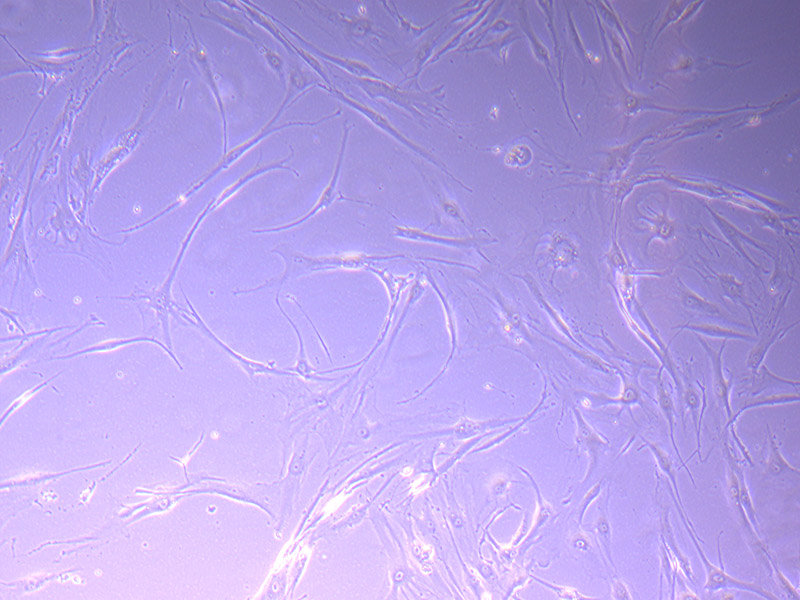

Brightfield microscope images of PHGL TB Cell Growth 20/09/21
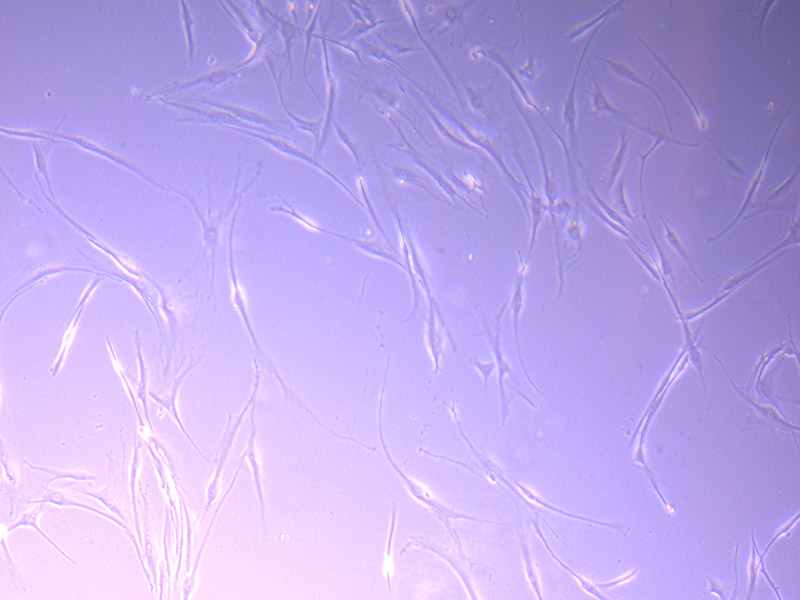
By 21/09/21 I decided there were enough cells to split into a second flask and freeze down one vial of cells. This will replenish the vial we used and bring our stock up to three vials.
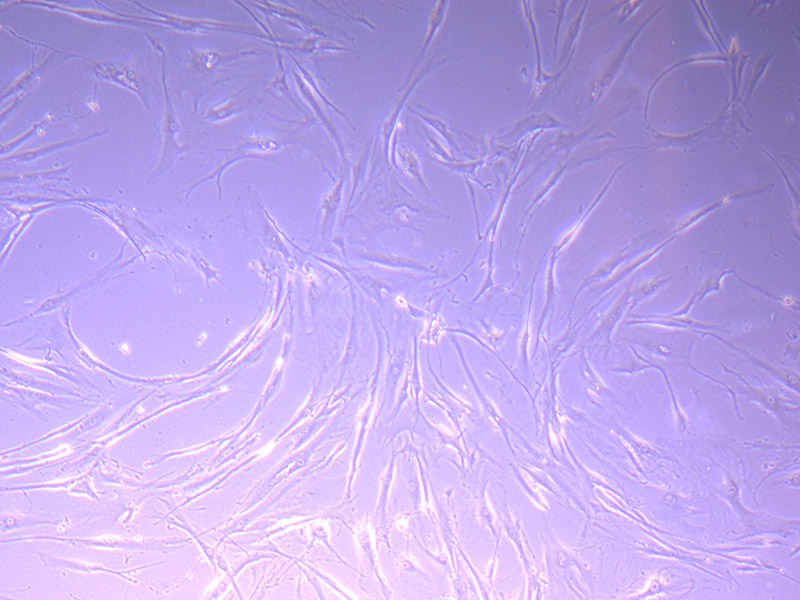
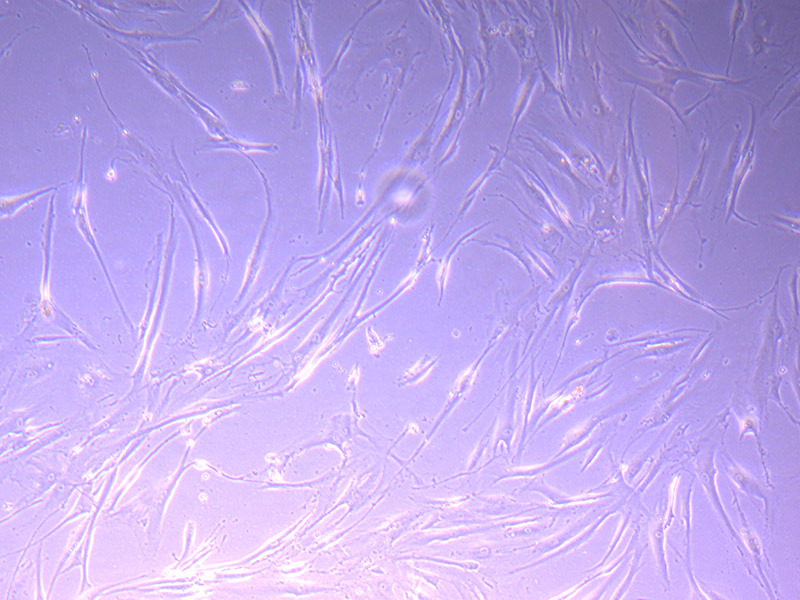
Brightfield microscope images of PHGL TB Cell Growth 21/09/21
Since the Stroke team mainly work with HBVPs, I reviewed standard protocols for fibroblasts to determine an optimum freezer mix. The recommendation from a number of sources is to include a higher rate of FBS at 30%, 10% DMSO (anti-free agent) and 70% media with a min. of 1 x 106 cells. I made up a total of 1.5mL freeze mix (including cells).
When I passaged the cells, I added 1mL new media to the cells solution. Since the cells were not 80 – 90% confluent, I decided to split them at a rate of 2/3. This means that the final freeze mix was: 150μL DMSO, 450μL FBS, 300μL media plus 600μL of cell mix. 1mL of this solution was added to cryovial and placed in a freeze box in the -80 degree freezer to be transferred into liquid nitrogen in the next day or so.
Since the cells were precious, I added the remaining cell freeze mix to a T25 flask with 5mL fresh media. There were also a few stubborn cells in the original T75 flask, (post passage), so I added 10mL new media to see if any of them might grow. Finally, the remaining 400μL cell mix (without freeze medium) to a new T75 flask with 10mL media.
At this point I had made up fresh DMEM media with the new batch of media, but decided to ‘wean’ the cells onto the new media at a 50/50 ratio of old to new. I am hoping that the new media will help the growth rate of the cells.