Logistics
I had a preliminary meeting with Dr Ash Mehta on Monday (21/2) to discuss the iPSC generation using blood. He uses the ThermoFisher CytoTune-iPS Sendai Reprogramming system and has generously offered to guide me through the cell reprogramming/iPSC generation timeline. The great thing about doing this is that the cells are technical immortal when in a stem cell state. This will enable me to achieve the primary project aims even if immortalisation of primary cells via SV40 does not work.
We followed up again on Tuesday (22/2) to go over the process in more detail and set up basic project requirements including blood collection in collaboration with the clinical research team. As part of this process, Ash introduced me to a lovely phlebotomist who agreed to collect my blood, as well as the Menzies Clinical Research Facility Manager to ensure everyone is informed about the project and correct processes are in place to move forward. After supplying project documentation and confirmation of ethics clearance, I received final sign off from the Chair o CRFMC to proceed with blood collection on 23/2/ – so full steam ahead!
Ash also showed me around his lab and allowed me to view the PMBCs (peripheral blood mononuclear cells) he thawed last week. The cells are cultured in suspension (non-adherent) and are circular in shape.
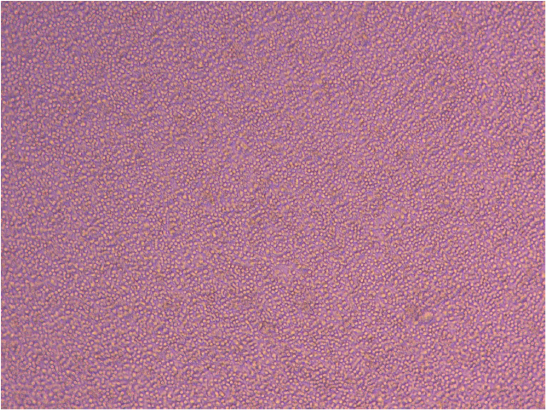
PBMCs in culture – image courtesy of Ashish Mehta
He also showed me some iPSCs and the difference between stem cell colonies and cells that have started to differentiate.
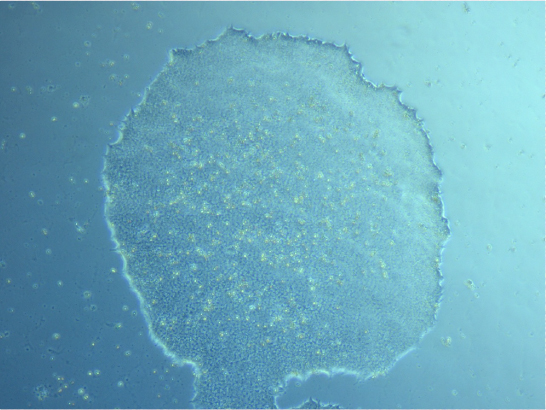
iPSC colony. Image courtesy of Ashish Mehta
About PMBCs
As part the introduction to cell reprogramming, Ash explained the basics and value of working with blood cells.
Blood is made from a number of different cell types including red blood cells (erythrocytes), white blood cells (macrophages, lymphocytes, monocytes, neutrophils, eosinophils, basophils ) and platelets (thrombocytes). Platelets and red blood cells have no nuclei so they cannot be reprogrammed and only the mononucleated (single nucleus) white blood cells are suitable for the process.
To isolate PMBCs, the blood undergoes gradient centrifugation which separates the blood into layers of cell types via density.
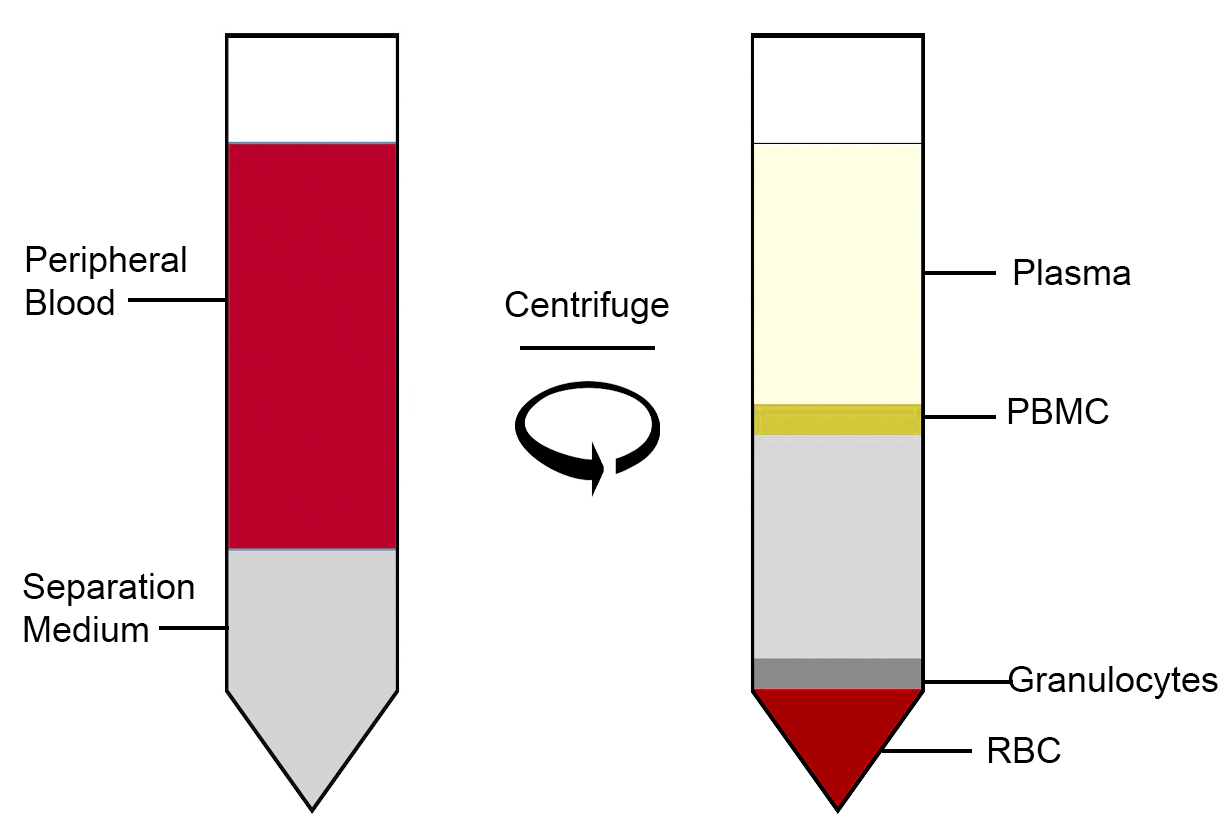
Diagram of peripheral blood separated into different layers including PBMCs ( round cells with a single nucleus: lymphocytes, monocytes, natural killer cells (NK cells), dendritic cells).
The advantage of using PBMCs is that you can tell more readily when the virus has initially successfully reprogrammed cells, as they change from non-adherent to adherent and start forming dense colonies of small cells.
The colonies need to be maintained meticulously as they tend to differentiate in culture (i.e. turn into (uncontrolled) specific cell types).
Ash indicated that when the blood is collected, it should be processed (PBMCs extracted) within a 4-hour window. A vial of blood should yield 4 – 5 million PBMCs, so he suggested that we freeze 4 x vials (1 x 106) as backup and proceed with a single 1 x 106 sample. This will also need careful planning to ensure that I am able to donate and process blood on the same day, plus move forward with the next steps involved.
On Thursday 24/2, Ash has kindly agreed for me to shadow him when he adds the Cytotune 2.0 (Sendai Virus reprogramming system) to the cultured PMBC samples. I’m looking forward to learning more.


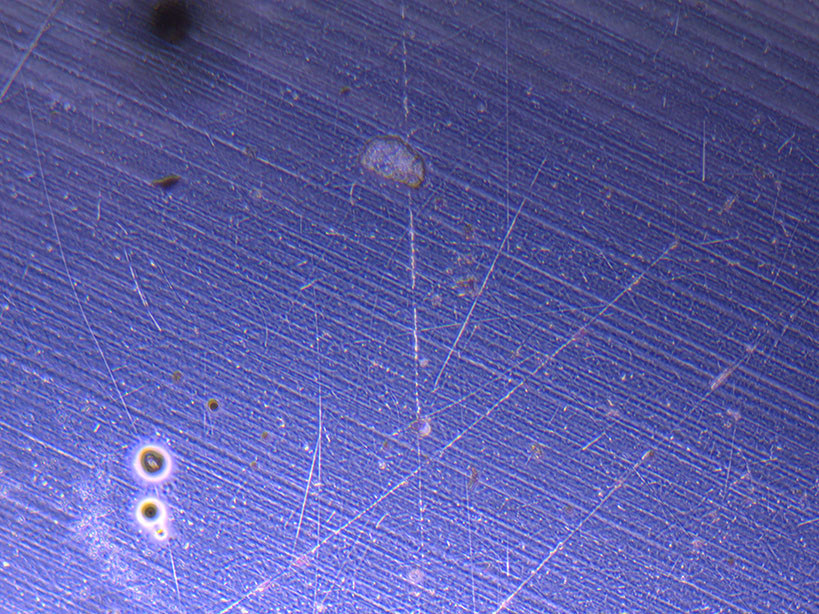
 Microscope images of cut glass dishes after H&E staining on 20/01/22 showing scratches on glass surface and no cells.
Microscope images of cut glass dishes after H&E staining on 20/01/22 showing scratches on glass surface and no cells. 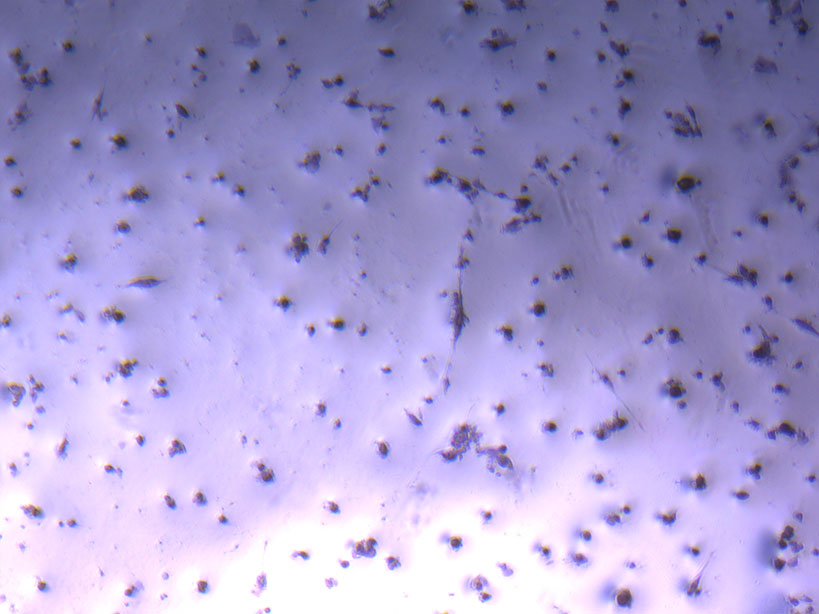 Microscope image of glass vial after H&E staining on 20/01/22. The image shows a vast number of dead cells that were not fixed in a live state.
Microscope image of glass vial after H&E staining on 20/01/22. The image shows a vast number of dead cells that were not fixed in a live state. 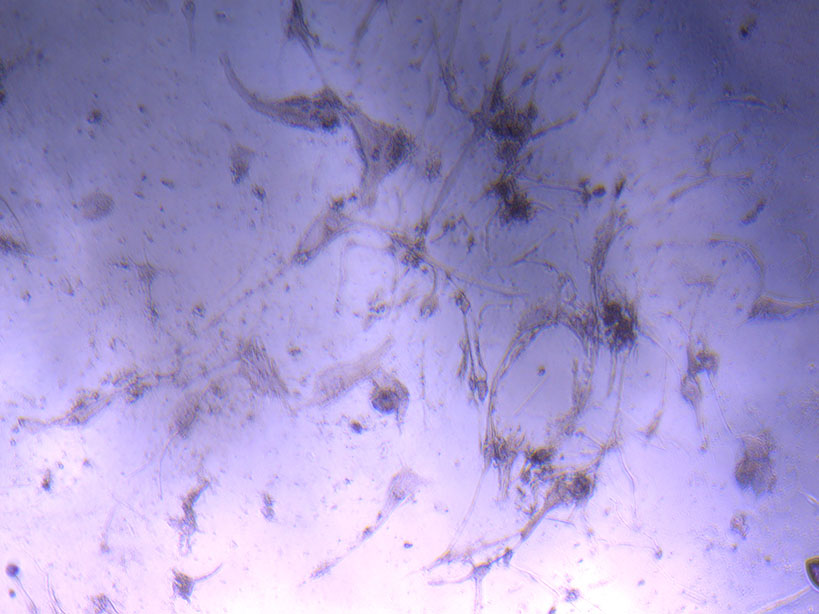 Microscope image of glass vial after H&E staining on 20/01/22. The image shows dried cell remnants.
Microscope image of glass vial after H&E staining on 20/01/22. The image shows dried cell remnants. 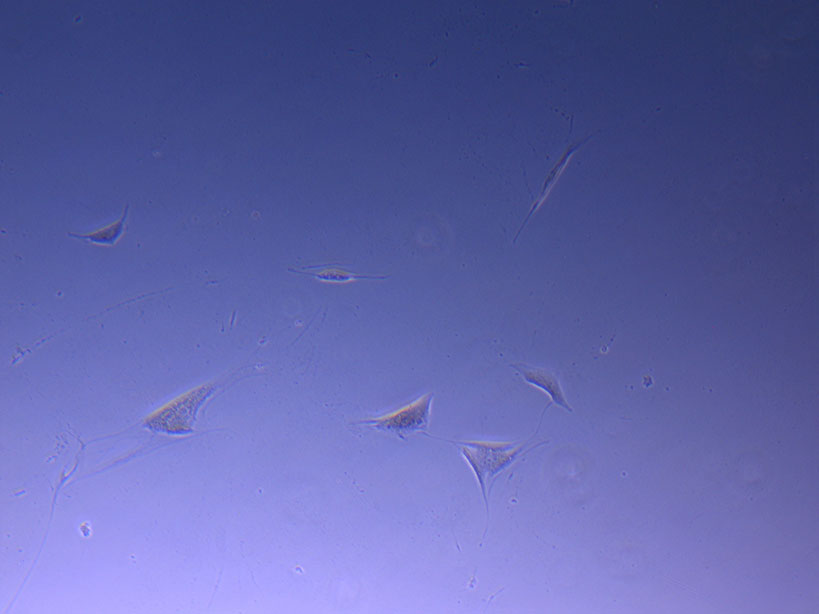
 Image of PHGL Tumour Baby cells Flask with original plating date (P 3, 07/10/21) and passage date (P4, 09/11/21) recorded.
Image of PHGL Tumour Baby cells Flask with original plating date (P 3, 07/10/21) and passage date (P4, 09/11/21) recorded. 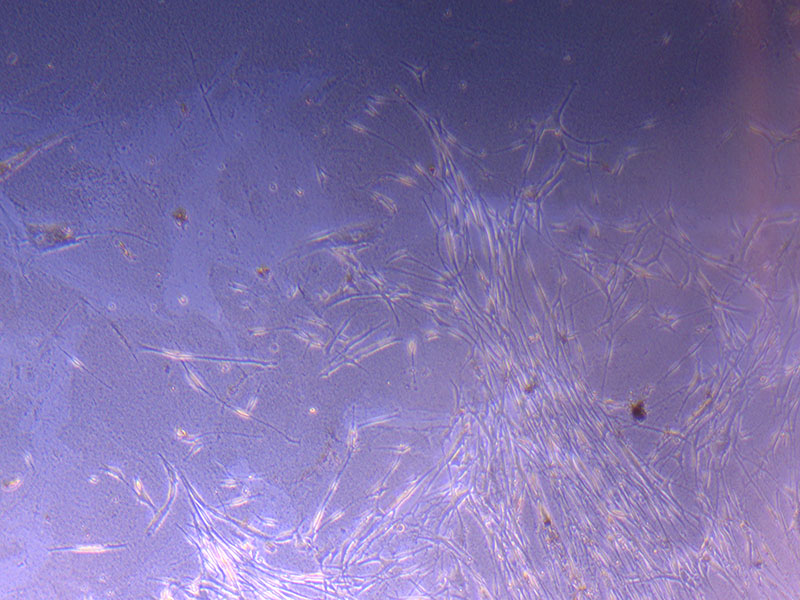 Light microscope image of PHGL Tumour Baby P3 in T75 flask [Org plated 07/10/21] imaged on 15/12/21.
Light microscope image of PHGL Tumour Baby P3 in T75 flask [Org plated 07/10/21] imaged on 15/12/21.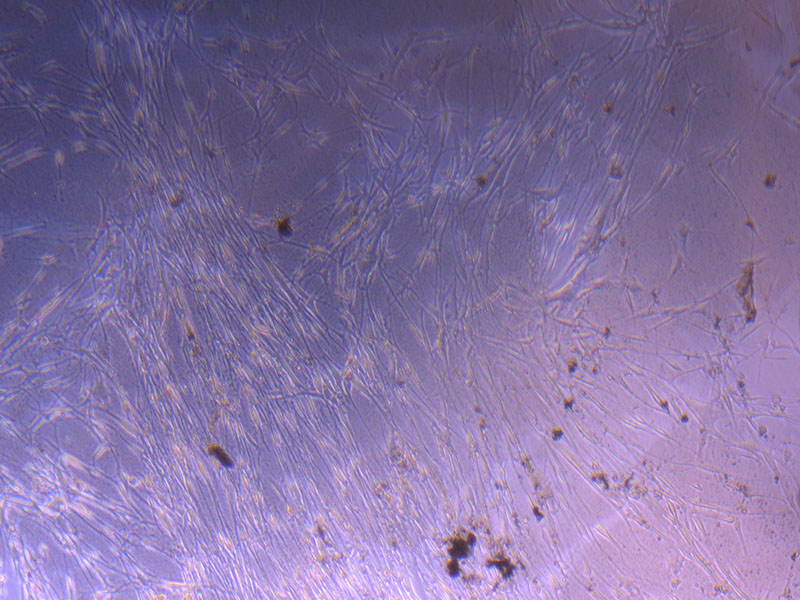
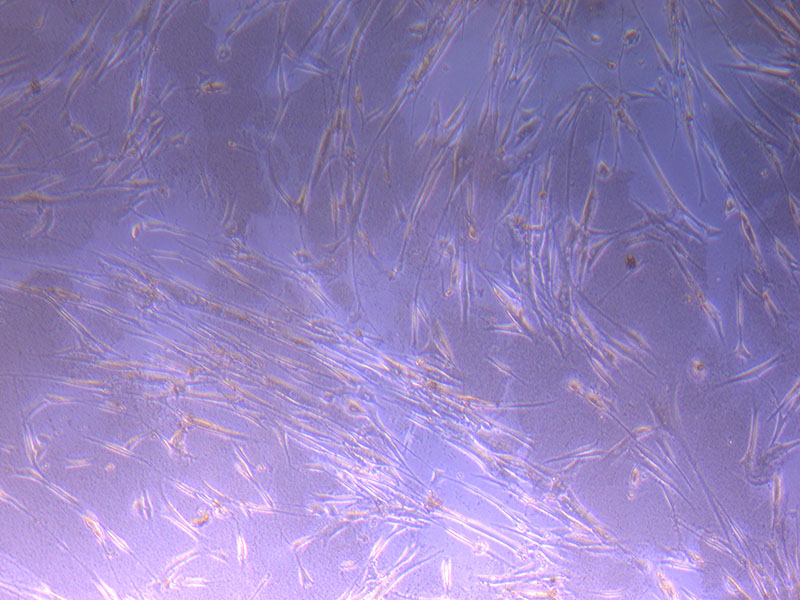
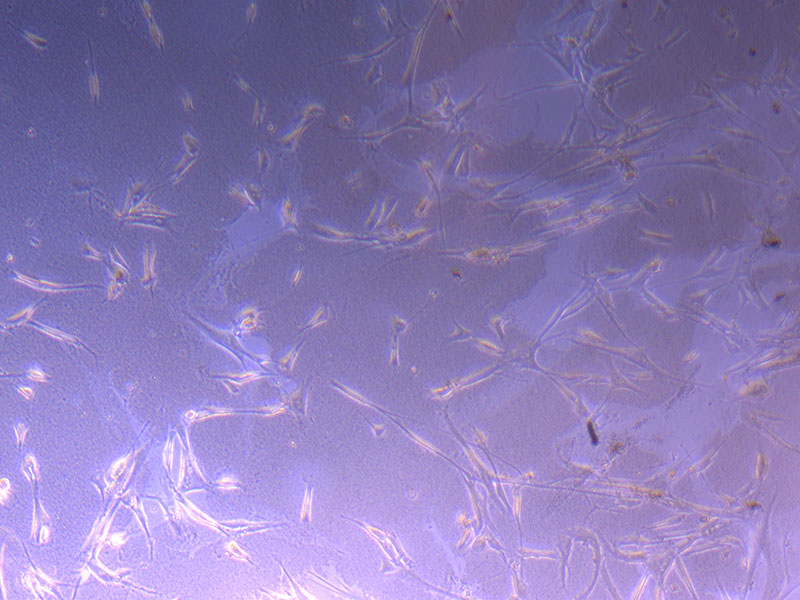
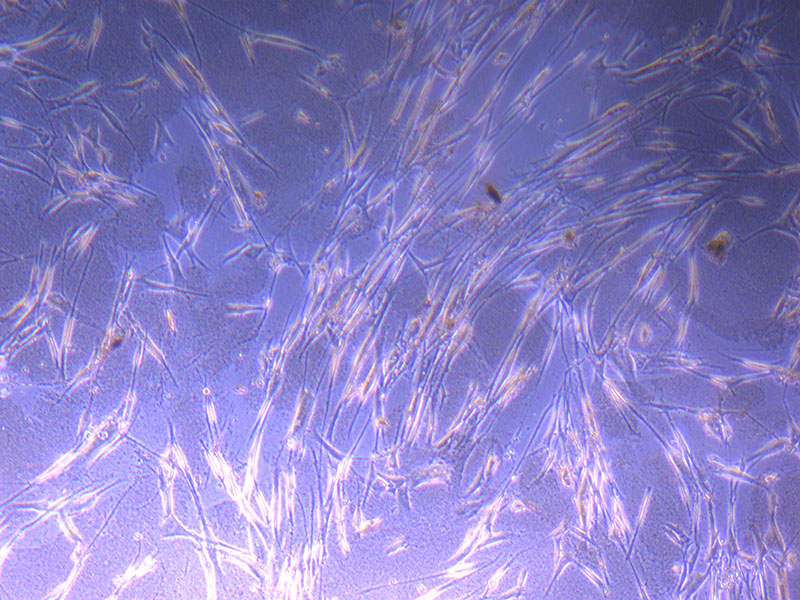 Light microscope images of PHGL Tumour Baby P4 in T75 flask [Org P3 plated 07/10/21 – passaged P4 on 9/11/21] imaged on 13/1/22 after cell maintenance.
Light microscope images of PHGL Tumour Baby P4 in T75 flask [Org P3 plated 07/10/21 – passaged P4 on 9/11/21] imaged on 13/1/22 after cell maintenance. 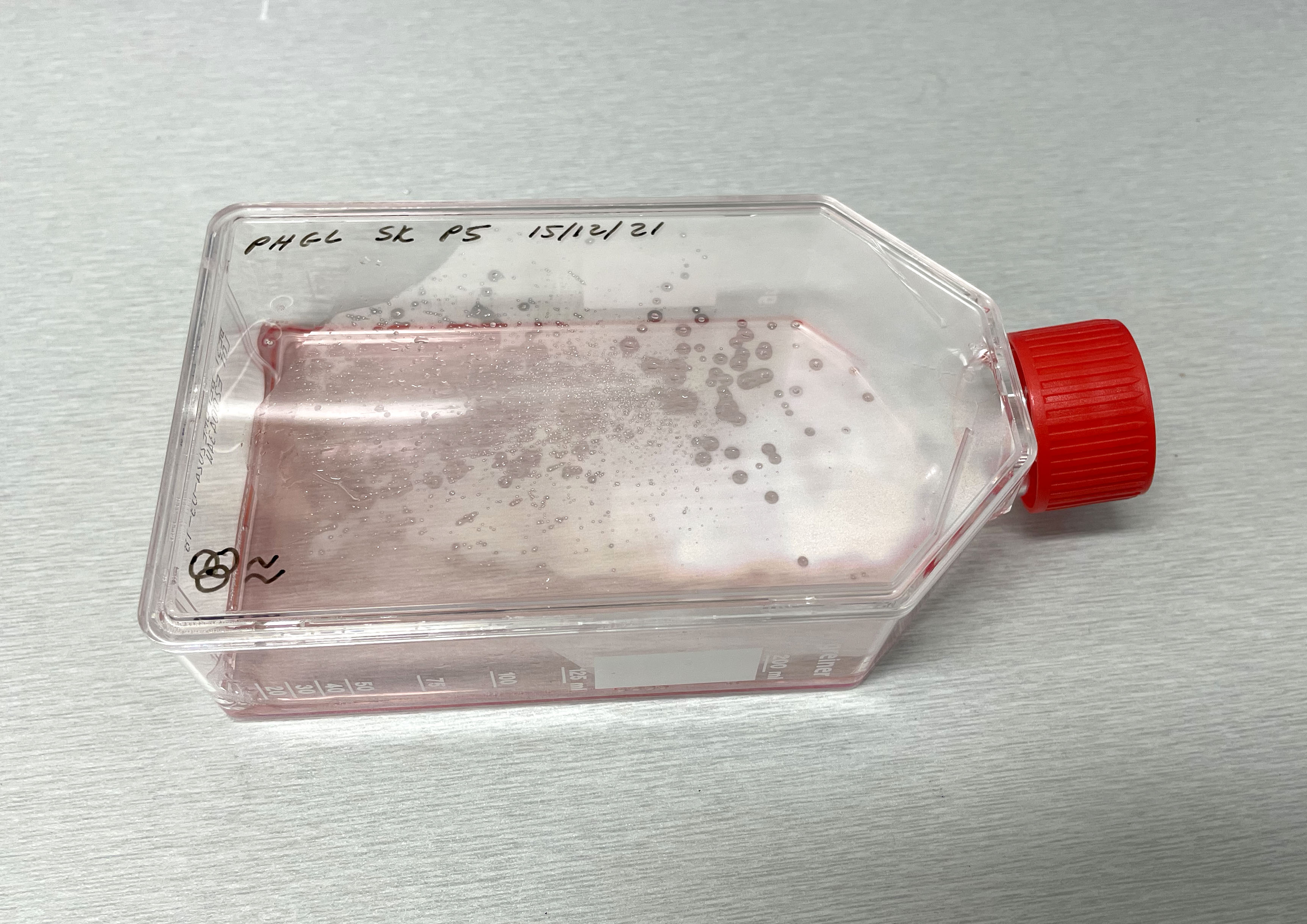 Photograph of PHGL Tumour Baby P5 in T75 flask plated on 15/12/21 prior to holidays.
Photograph of PHGL Tumour Baby P5 in T75 flask plated on 15/12/21 prior to holidays. 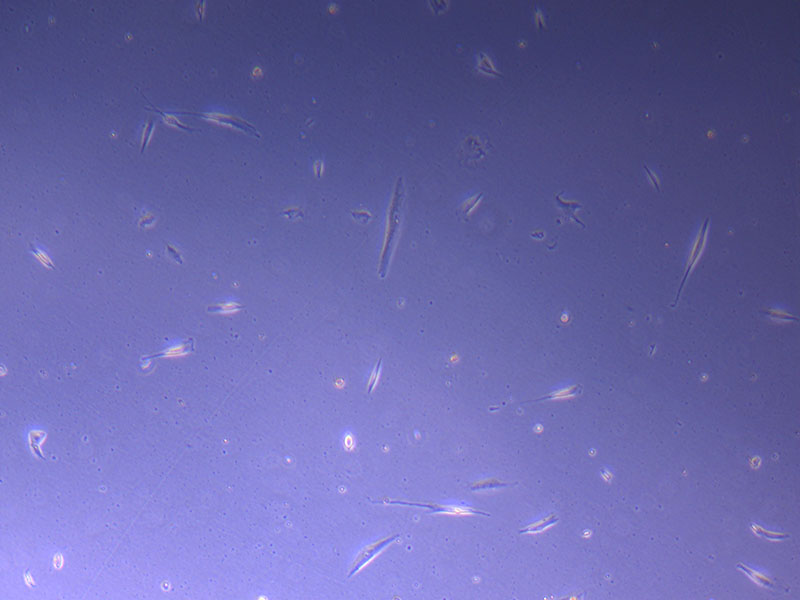
 Light microscope image of PHGL Tumour Baby P5 in T75 flask plated on 15/12/21.
Light microscope image of PHGL Tumour Baby P5 in T75 flask plated on 15/12/21.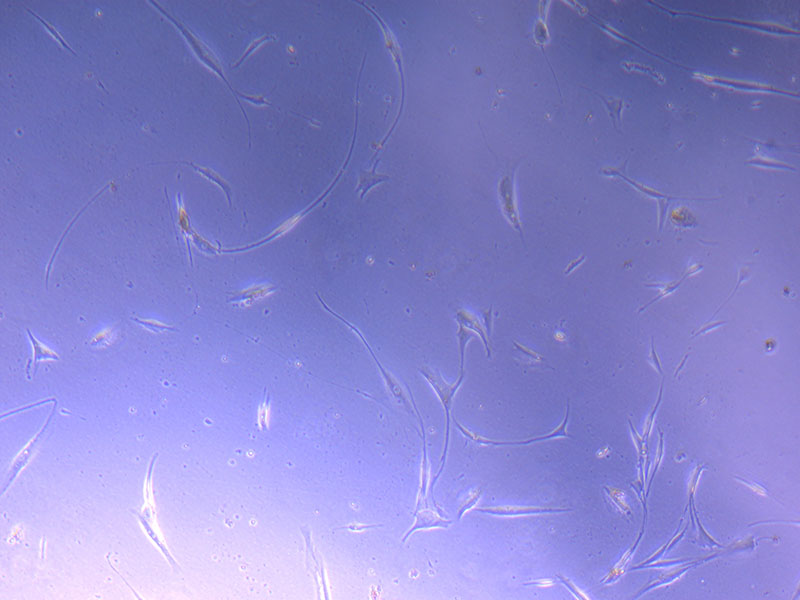
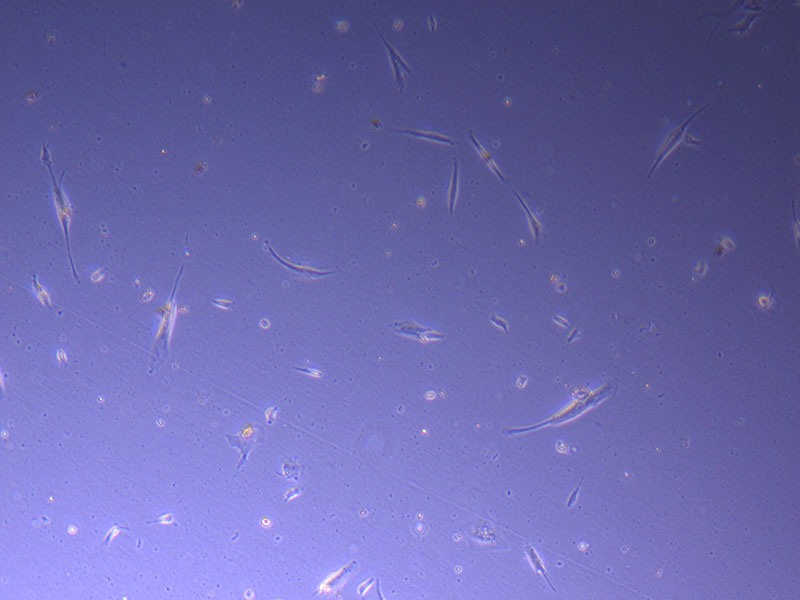
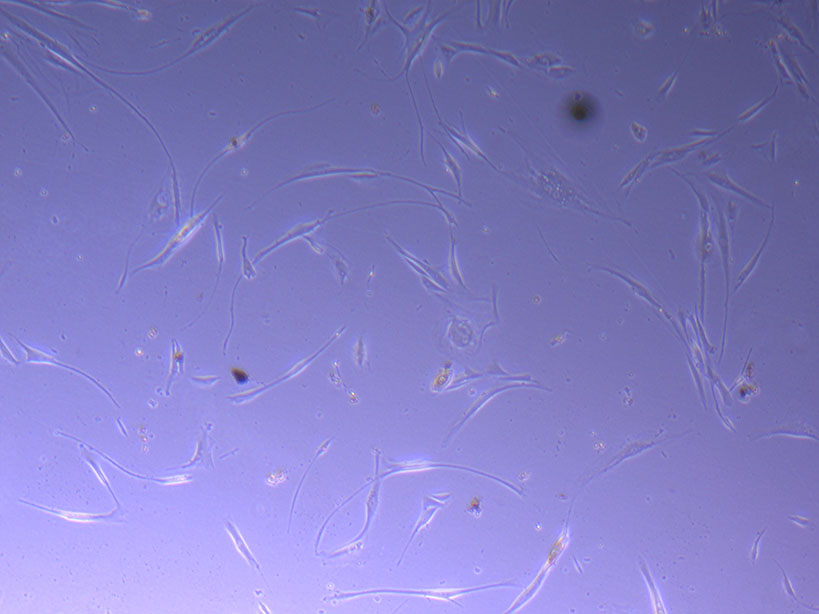 Light microscope images of PHGL Tumour Baby P5 in T75 flask plated on 15/12/21 and imaged on 13/1/22.
Light microscope images of PHGL Tumour Baby P5 in T75 flask plated on 15/12/21 and imaged on 13/1/22.  Photograph of PHGL Tumour Baby P5 in T75 flask #2 plated on 15/12/21.
Photograph of PHGL Tumour Baby P5 in T75 flask #2 plated on 15/12/21.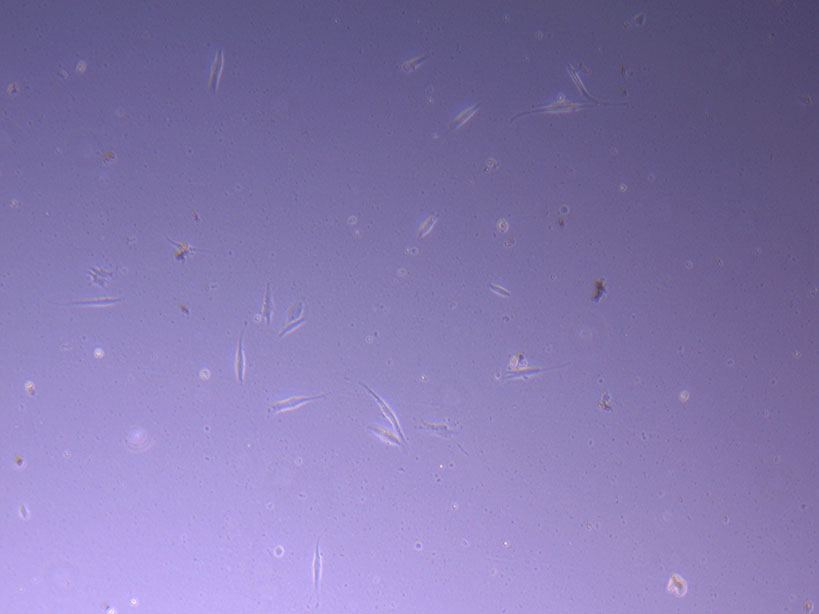 Light microscope image of PHGL Tumour Baby P5 in T75 flask 2 plated on 15/12/21.
Light microscope image of PHGL Tumour Baby P5 in T75 flask 2 plated on 15/12/21.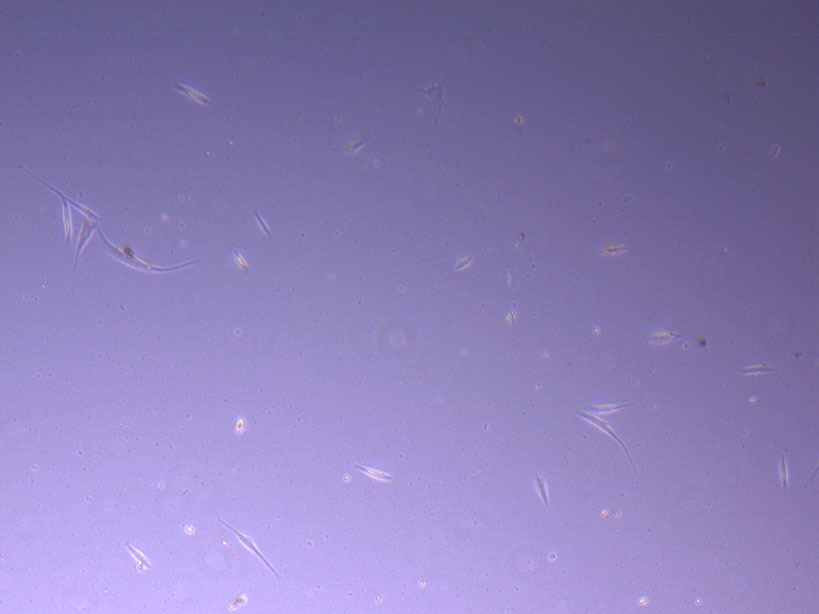 Light microscope image of PHGL Tumour Baby P5 in T75 flask 2 plated on 15/12/21.
Light microscope image of PHGL Tumour Baby P5 in T75 flask 2 plated on 15/12/21.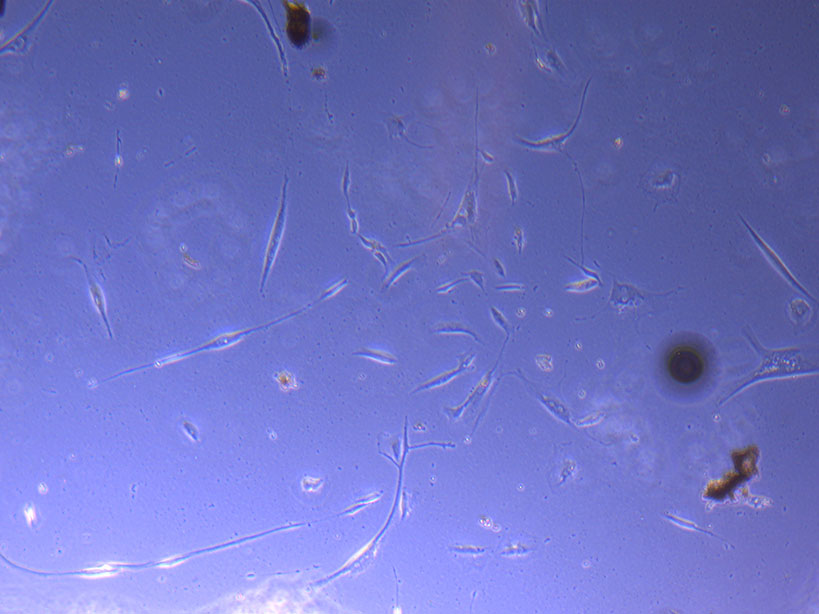
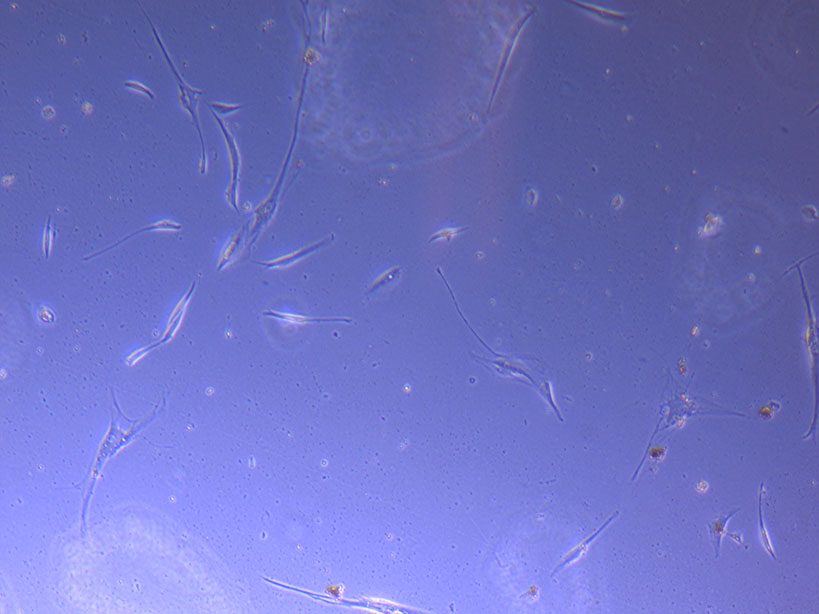 Light microscope image of PHGL Tumour Baby P5 in T75 flask 2 imaged on 13/1/22.
Light microscope image of PHGL Tumour Baby P5 in T75 flask 2 imaged on 13/1/22.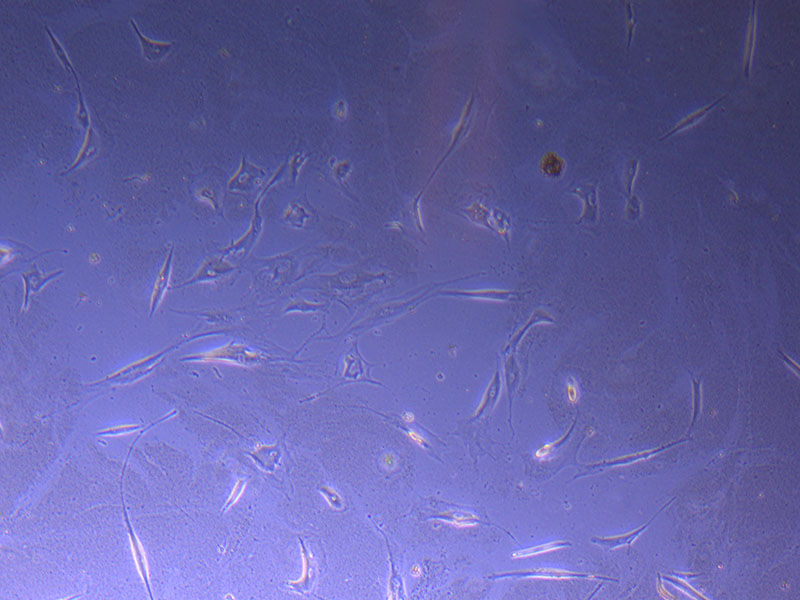 Light microscope image of PHGL TB cells at 13/1/22 showing a main cluster area of remaining cells.
Light microscope image of PHGL TB cells at 13/1/22 showing a main cluster area of remaining cells.  Photograph of P1 PHGL TB flask originally plated out by Jo-Maree on 10/0921.
Photograph of P1 PHGL TB flask originally plated out by Jo-Maree on 10/0921. 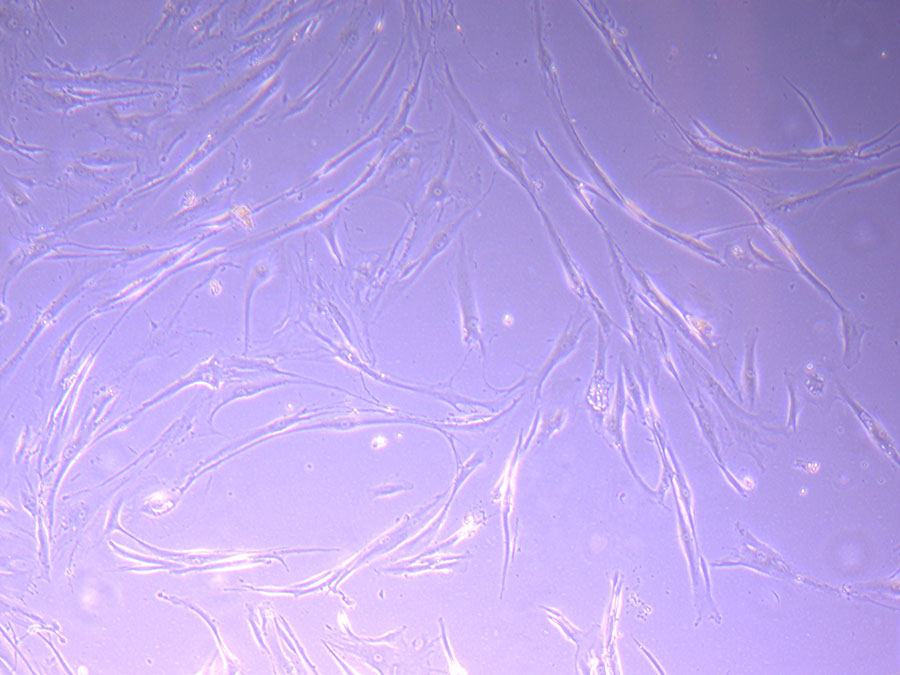 Light microscope image of P1 PHGL TB cells taken on 21/09/21 prior to passage.
Light microscope image of P1 PHGL TB cells taken on 21/09/21 prior to passage. 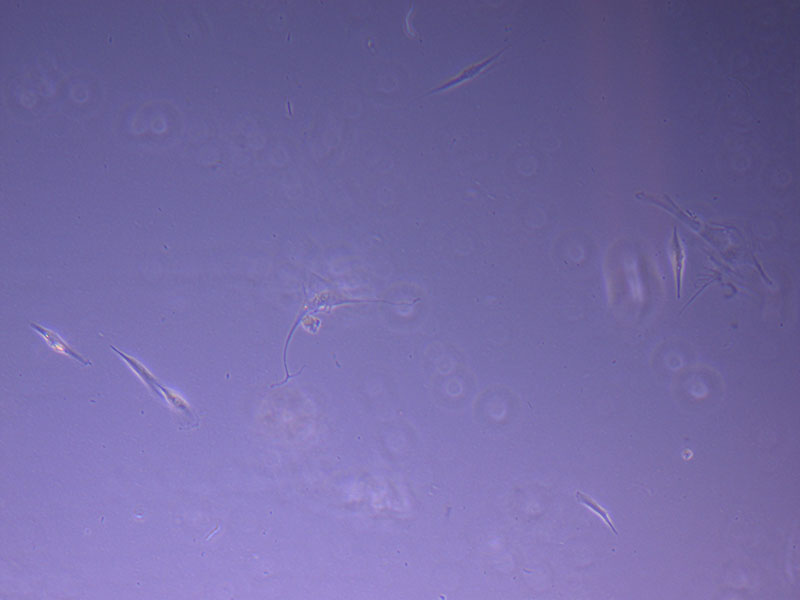 Light microscope image of P2 PHGL TB cells (in original flask) taken on 23/09/21 after cell passage on 21/09/21. A few cells remain visible in the flask.
Light microscope image of P2 PHGL TB cells (in original flask) taken on 23/09/21 after cell passage on 21/09/21. A few cells remain visible in the flask. 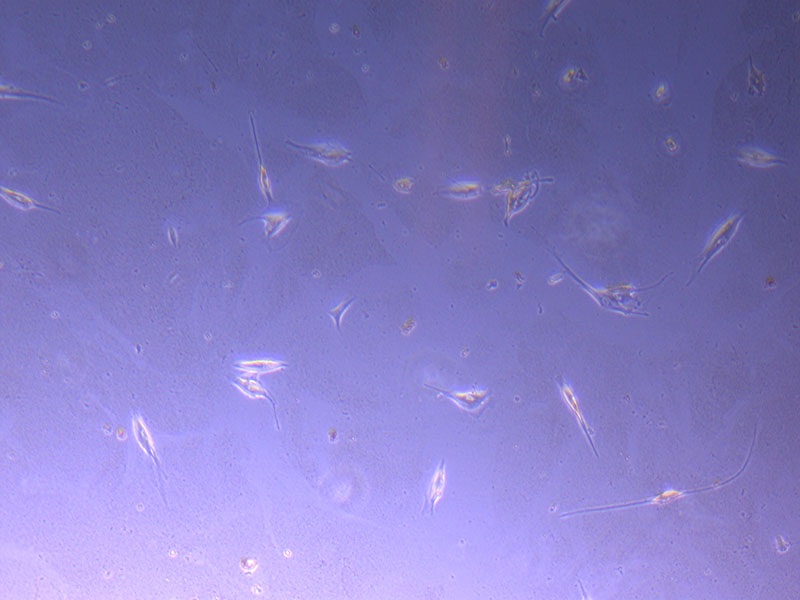 Light microscope image of P2 PHGL TB cells (in original flask) taken on 13/1/22. The image reveals a ‘sprinkle’ of cells beyond the main cluster.
Light microscope image of P2 PHGL TB cells (in original flask) taken on 13/1/22. The image reveals a ‘sprinkle’ of cells beyond the main cluster. 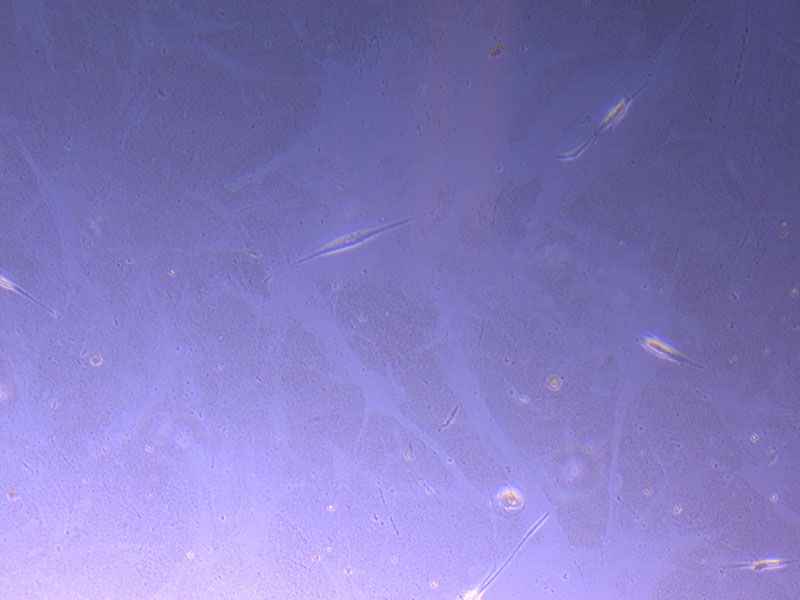
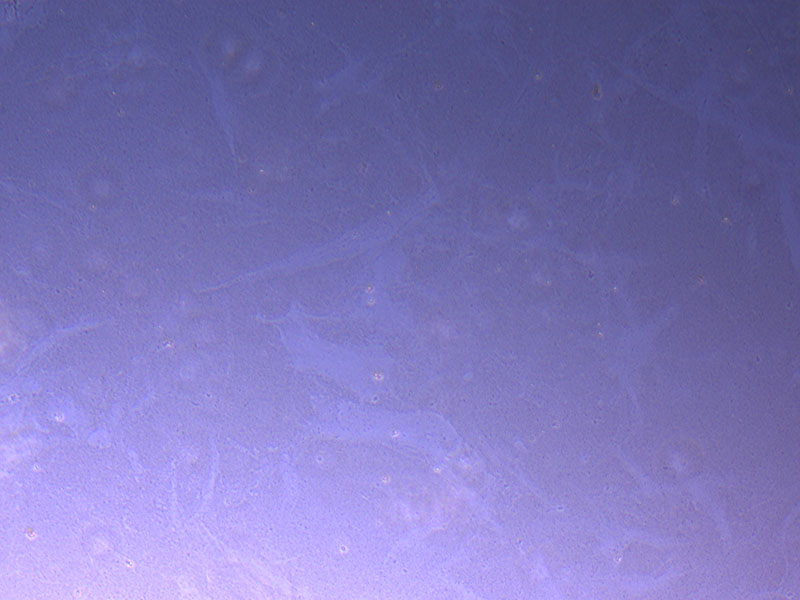 Light microscope images of P2 PHGL TB cells (in original flask) taken on 13/1/22. The image reveals trails of cellular movement and existence.
Light microscope images of P2 PHGL TB cells (in original flask) taken on 13/1/22. The image reveals trails of cellular movement and existence. 
 Unwrapping Petri dishes and getting ready to coat culture glassware with PLL.
Unwrapping Petri dishes and getting ready to coat culture glassware with PLL.  PLL coated glassware in Petri dishes ready for incubation.
PLL coated glassware in Petri dishes ready for incubation. 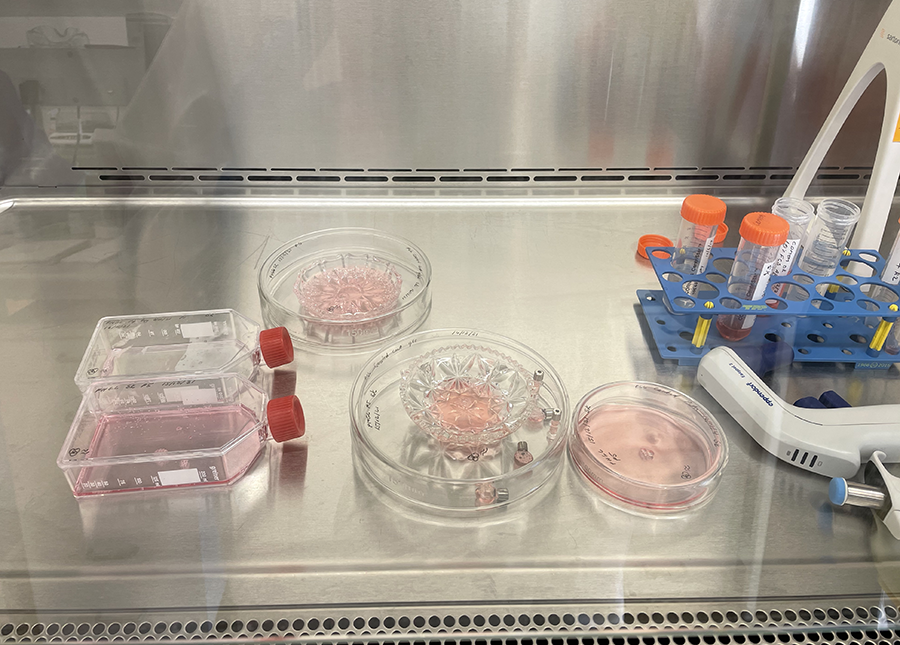 Cut glass vessels with cells ready for incubation.
Cut glass vessels with cells ready for incubation.  Clear cut glass dish – approx 12cm diameter with central star motif and radiating pattern.
Clear cut glass dish – approx 12cm diameter with central star motif and radiating pattern. Cut glass dish and wrapped dish ready for sterilisation.
Cut glass dish and wrapped dish ready for sterilisation. Selection of glass vials for cell culture trial including metal closures.
Selection of glass vials for cell culture trial including metal closures.  Glass vessels in autoclave bags ready for sterilisation.
Glass vessels in autoclave bags ready for sterilisation.  Petri dishes and other glass items in autoclave bags ready for sterilisation.
Petri dishes and other glass items in autoclave bags ready for sterilisation. 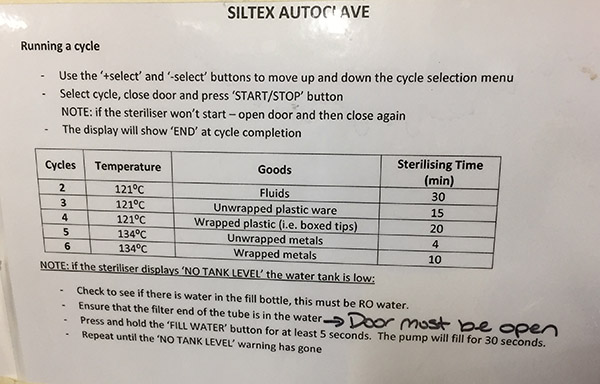 Autoclave instructions with cycle details.
Autoclave instructions with cycle details.  Autoclaved bags containing sterilised Petri dishes.
Autoclaved bags containing sterilised Petri dishes.  Autoclaved bags stored in lab area, ready for use.
Autoclaved bags stored in lab area, ready for use.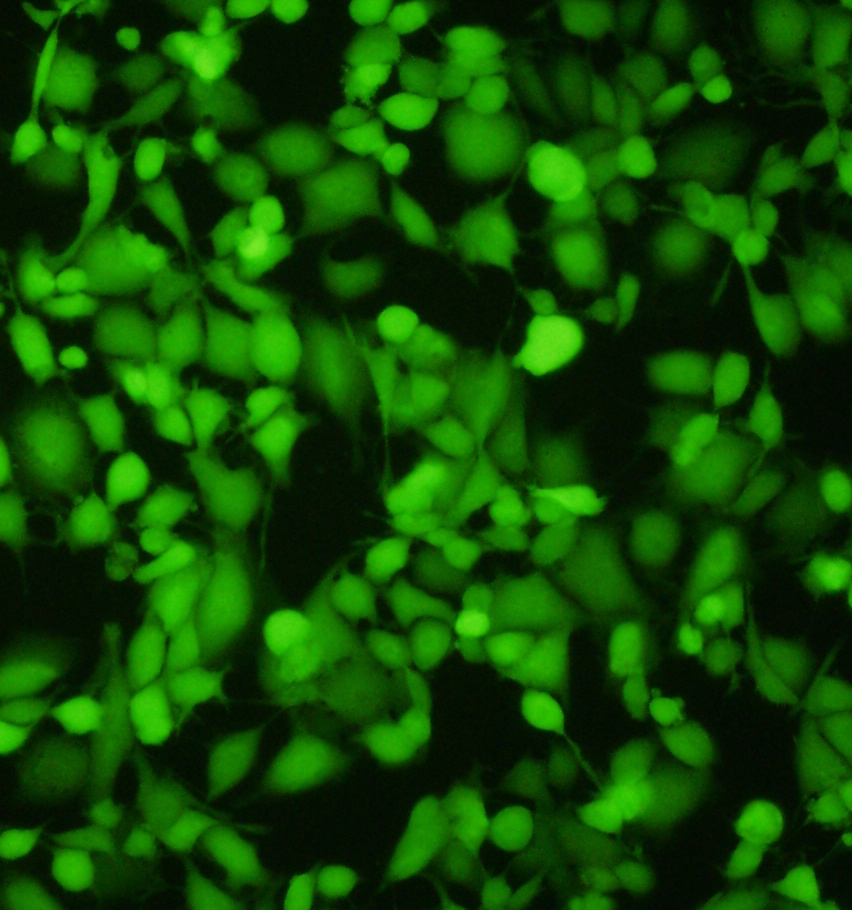 Impressive image of Calcein dye – live cells fluoresce a vibrant green – image via
Impressive image of Calcein dye – live cells fluoresce a vibrant green – image via 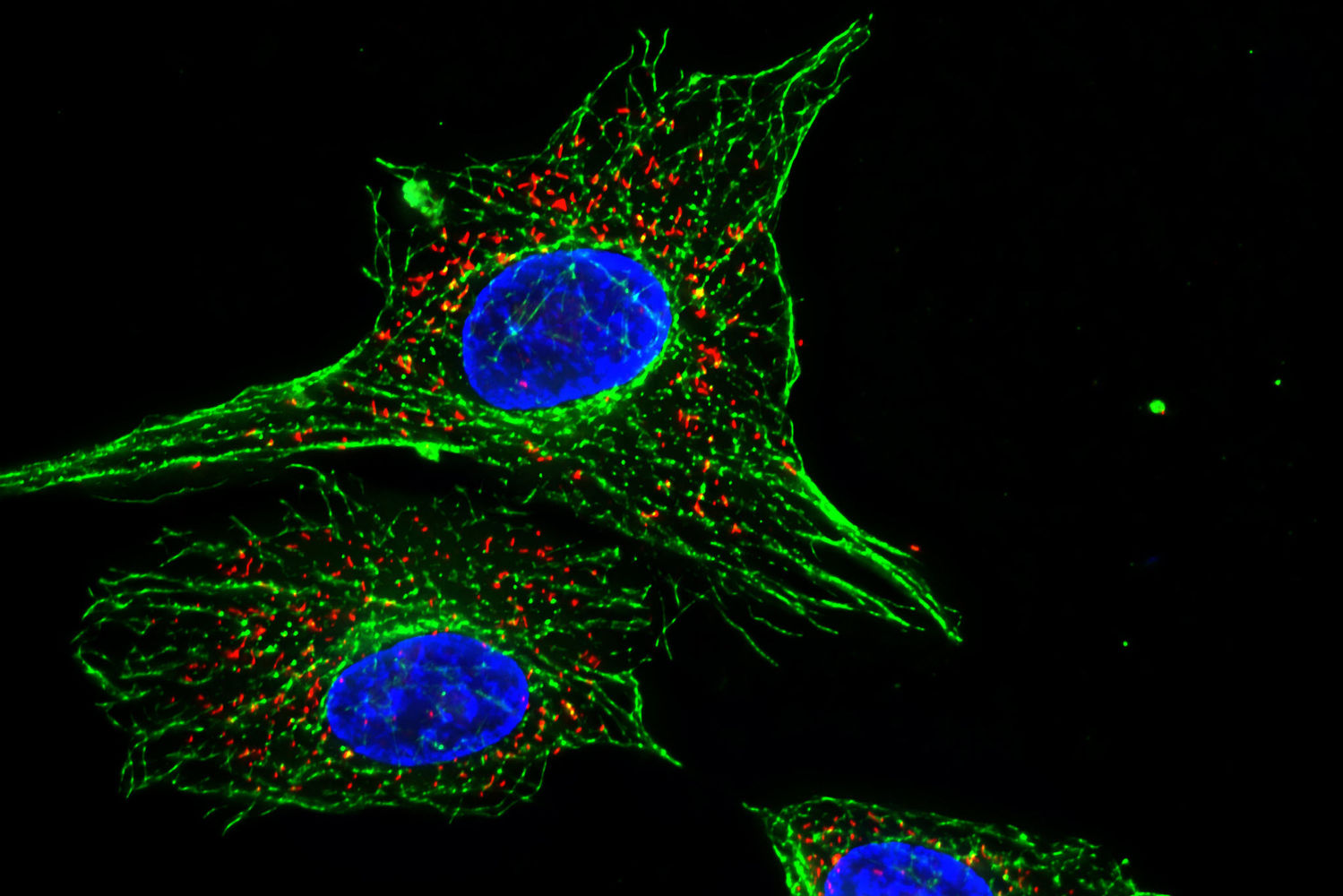 Image of fluorescent cells via
Image of fluorescent cells via  Basic H&E staining protocol from Jo-Maree. We only need to follow the staining process.
Basic H&E staining protocol from Jo-Maree. We only need to follow the staining process. Petri Dish on bench in Histology Lab at MSP with Erlenmeyer flask containing distilled water for washing.
Petri Dish on bench in Histology Lab at MSP with Erlenmeyer flask containing distilled water for washing.  Hematoxylin Stain – deep red stain
Hematoxylin Stain – deep red stain 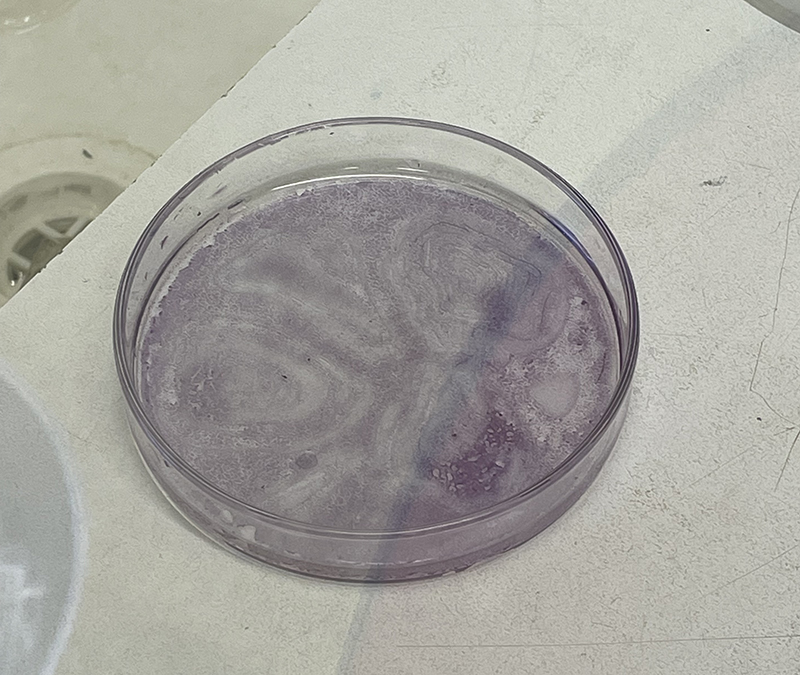

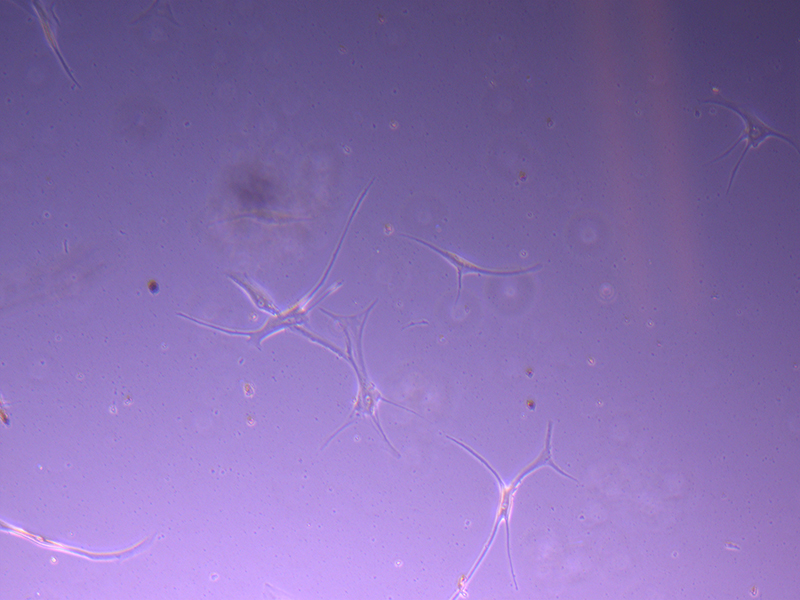 T25 – Flask 1 P 3, 11/10/21
T25 – Flask 1 P 3, 11/10/21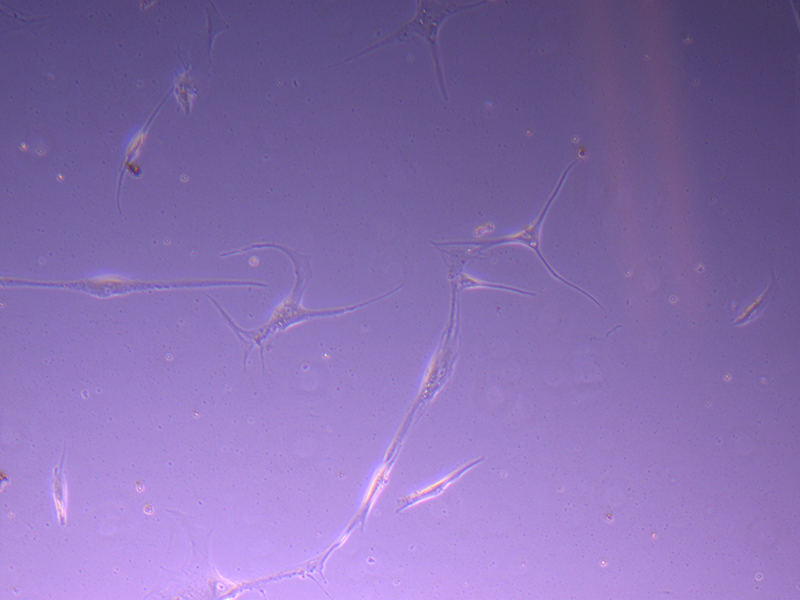 T25 – Flask 1 P 3, 11/10/21
T25 – Flask 1 P 3, 11/10/21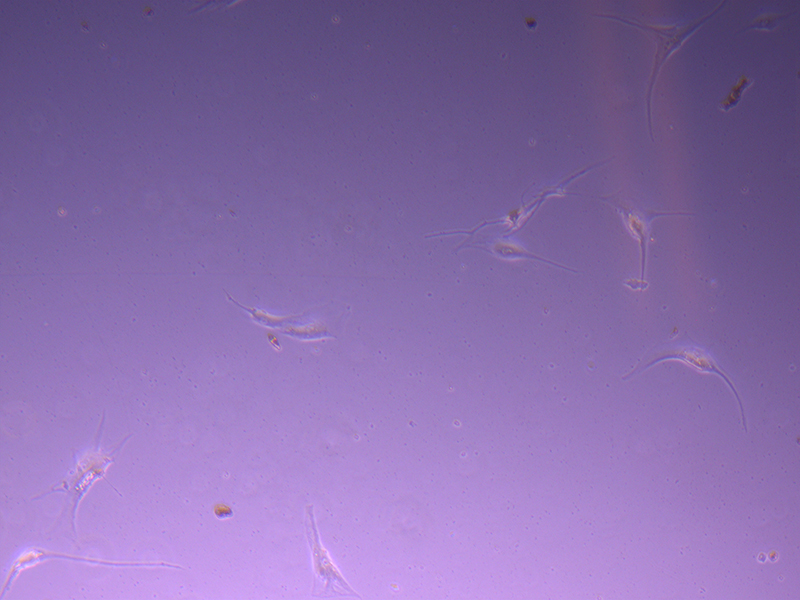 T25 – Flask 2 P 3, 11/10/21
T25 – Flask 2 P 3, 11/10/21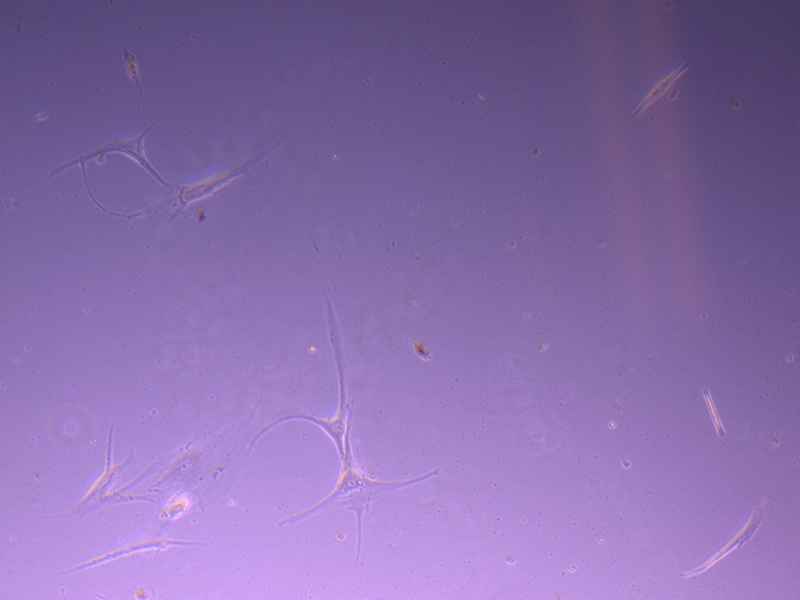 T25 – Flask 2 P 3, 11/10/21
T25 – Flask 2 P 3, 11/10/21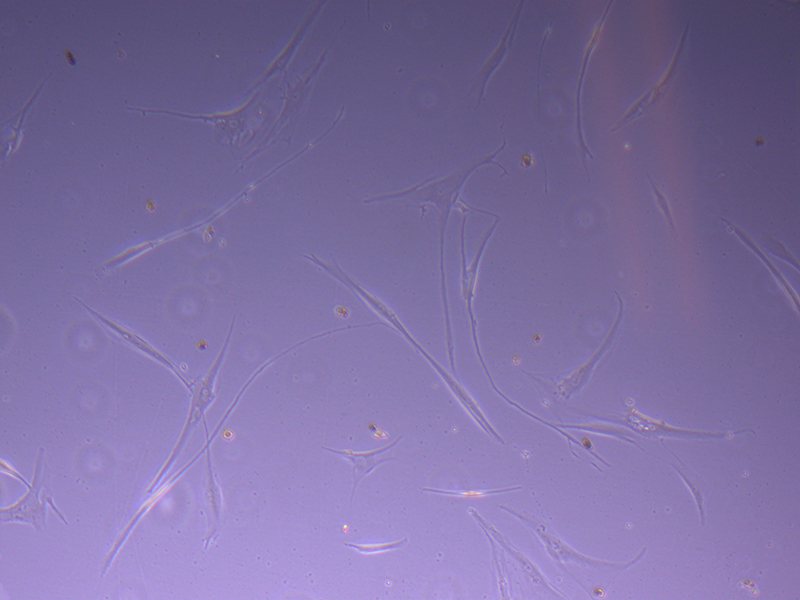 T75 – Flask 1 P 3, 11/10/21
T75 – Flask 1 P 3, 11/10/21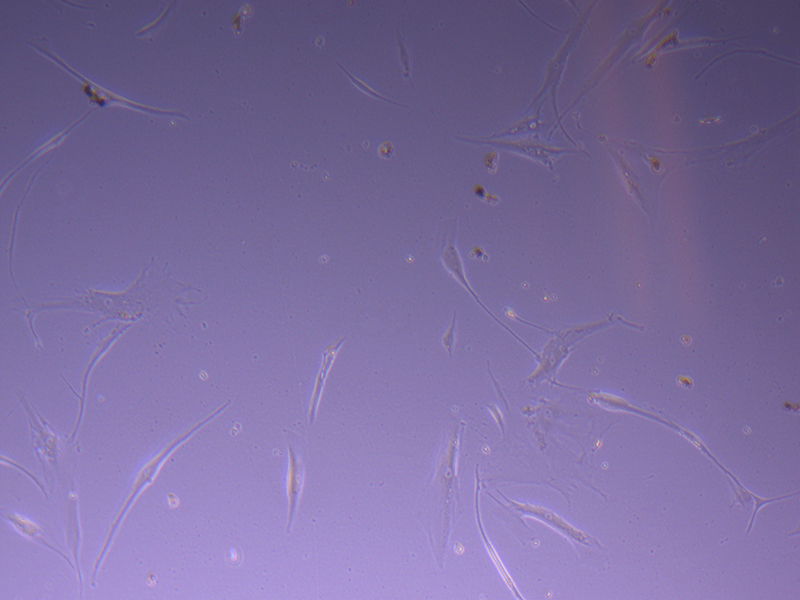 T75 – Flask 1 P 3, 11/10/21
T75 – Flask 1 P 3, 11/10/21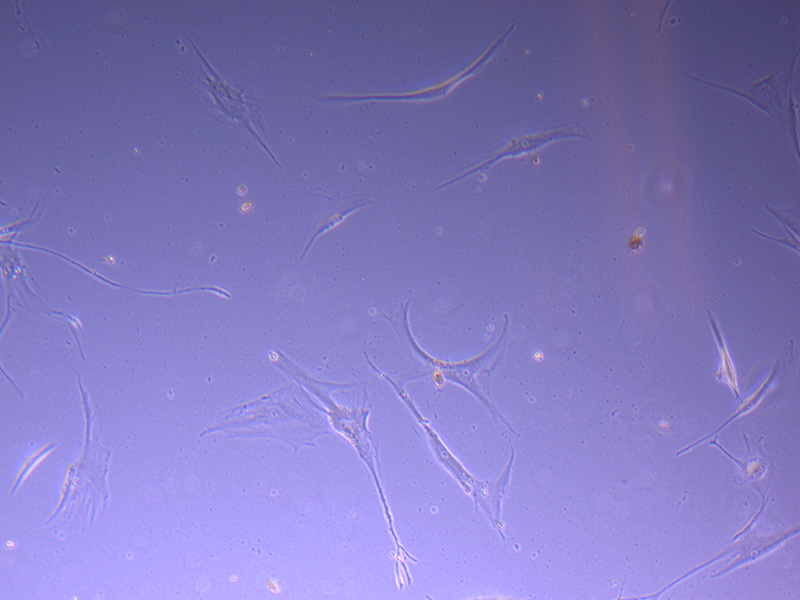 T75 – Flask 2 P 3, 11/10/21
T75 – Flask 2 P 3, 11/10/21 T75 – Flask 2 P 3, 11/10/21
T75 – Flask 2 P 3, 11/10/21