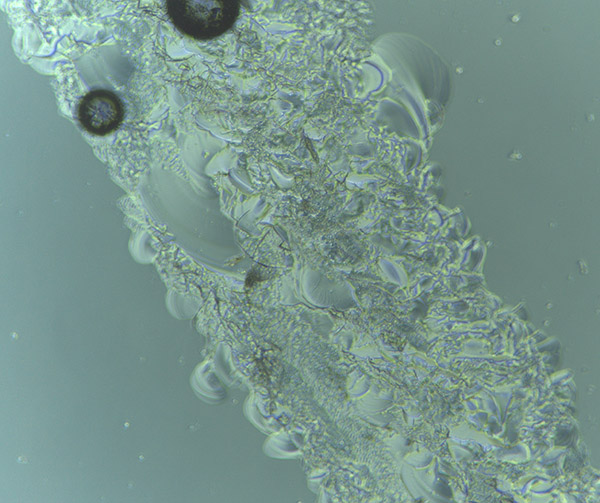
Following more detailed review of cell flasks, there are some hardy ‘survivors’ of my lab-free holiday.
 Light microscope image of cut glass dish with media containing dead cell debris and evidence of a small number of surviving cells.
Light microscope image of cut glass dish with media containing dead cell debris and evidence of a small number of surviving cells.
After removing the old media, it was easier to see the remaining cells:
Light microscope image of cut glass dish in PBS showing evidence of a small number of surviving cells.
This image shows even more evidence of cell survival:
 Light microscope image of cut glass dish in PBS showing further evidence of a small number of surviving cells.
Light microscope image of cut glass dish in PBS showing further evidence of a small number of surviving cells.
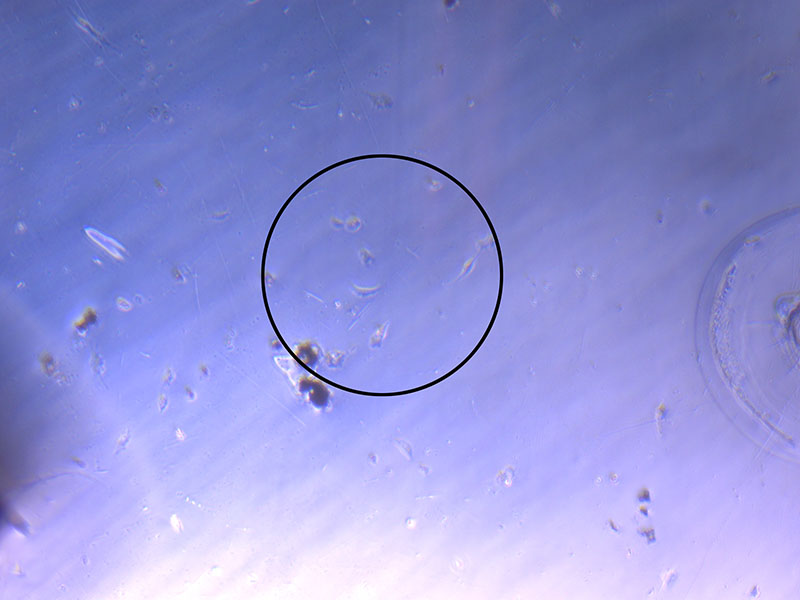
 Light microscope image of cut glass dish in PBS with likely cells circled. There are a few additional potential cells visible, but I have only circled the most obvious.
Light microscope image of cut glass dish in PBS with likely cells circled. There are a few additional potential cells visible, but I have only circled the most obvious.
To get an even better sense of survivors, I will fix (in 4% PFA) and H&E stain 2/3 of the cut glass dishes. The flatter cut glass dish and Petri dish (with more potential for cell survival will be maintained in the incubator to see how they fare over the next week).
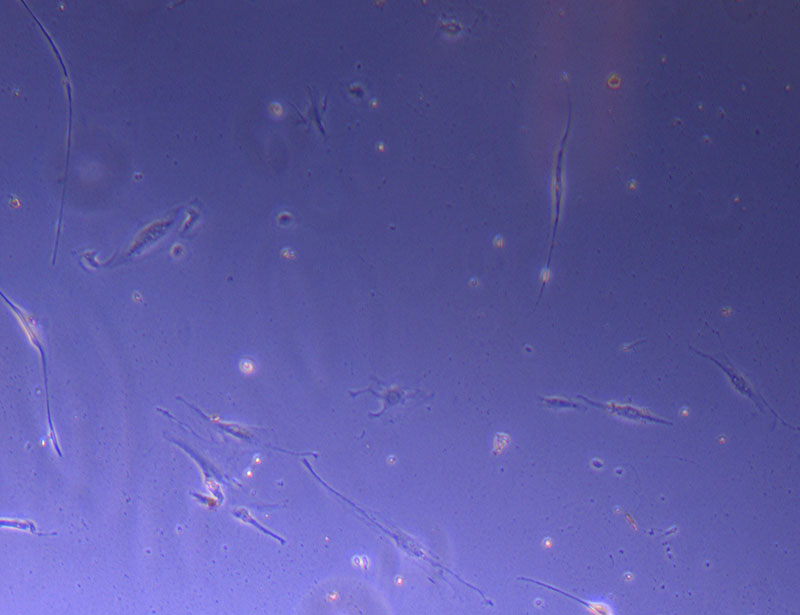 Light microscope image of Petri dish with fresh complete media with live cells.
Light microscope image of Petri dish with fresh complete media with live cells.
I will also fix and stain the glass vessels. It is less likely that these will yield anything interesting, but it will help me troubleshoot how to do the protocol with the tiny openings – it is very difficult to effectively remove the media – even with a 20ul pipette and tip 🙁

 Unwrapping Petri dishes and getting ready to coat culture glassware with PLL.
Unwrapping Petri dishes and getting ready to coat culture glassware with PLL.  PLL coated glassware in Petri dishes ready for incubation.
PLL coated glassware in Petri dishes ready for incubation. 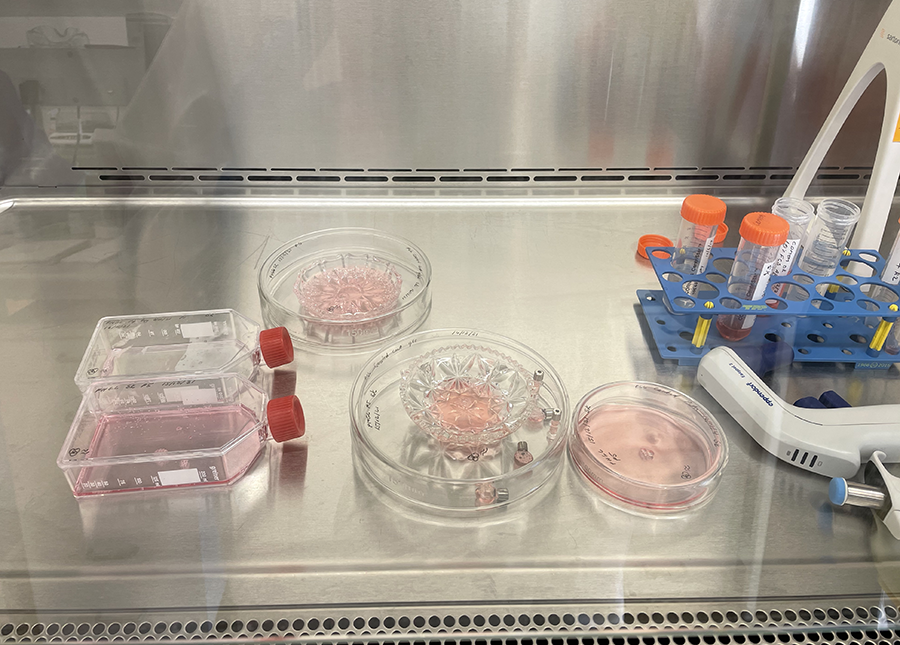 Cut glass vessels with cells ready for incubation.
Cut glass vessels with cells ready for incubation.  Basic H&E staining protocol from Jo-Maree. We only need to follow the staining process.
Basic H&E staining protocol from Jo-Maree. We only need to follow the staining process. Petri Dish on bench in Histology Lab at MSP with Erlenmeyer flask containing distilled water for washing.
Petri Dish on bench in Histology Lab at MSP with Erlenmeyer flask containing distilled water for washing.  Hematoxylin Stain – deep red stain
Hematoxylin Stain – deep red stain 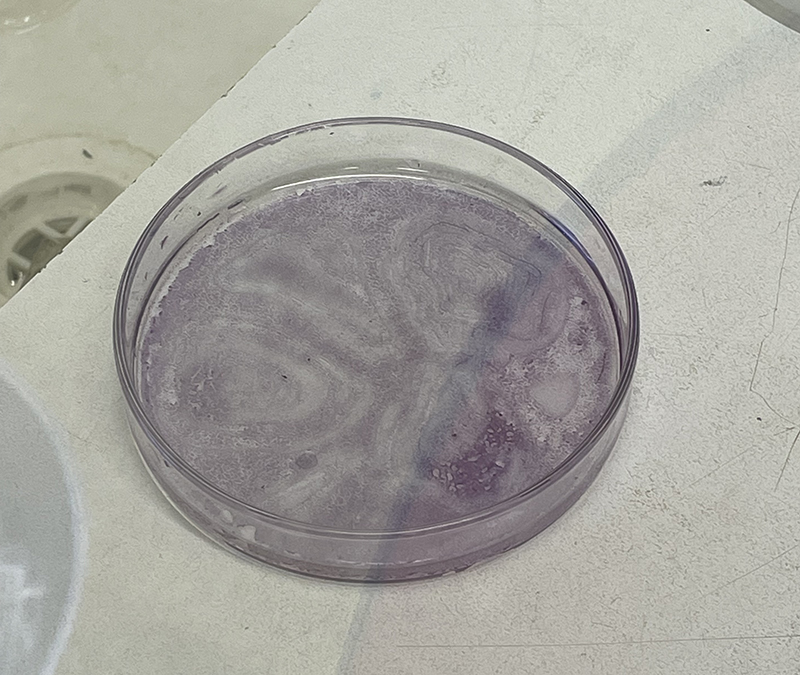




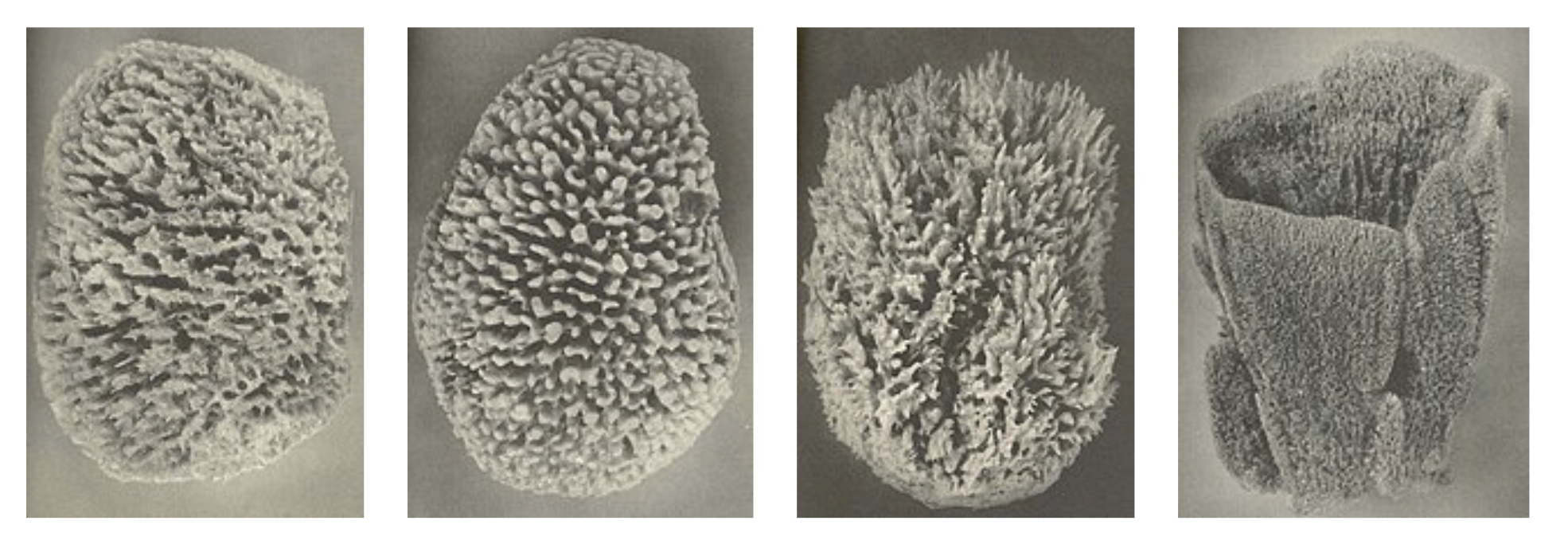 Different sponges from
Different sponges from 


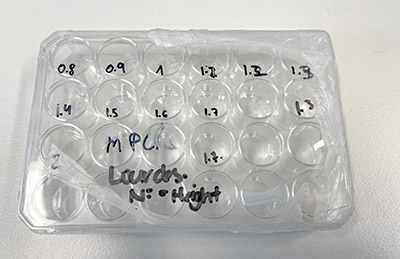 MPCL Scaffolds – tubular structures at varying heights.
MPCL Scaffolds – tubular structures at varying heights. Scaffolds in Petri Dish – flat square structures.
Scaffolds in Petri Dish – flat square structures.  MPCL+CaP coating
MPCL+CaP coating MPCL+CaP coating – tube structures.
MPCL+CaP coating – tube structures.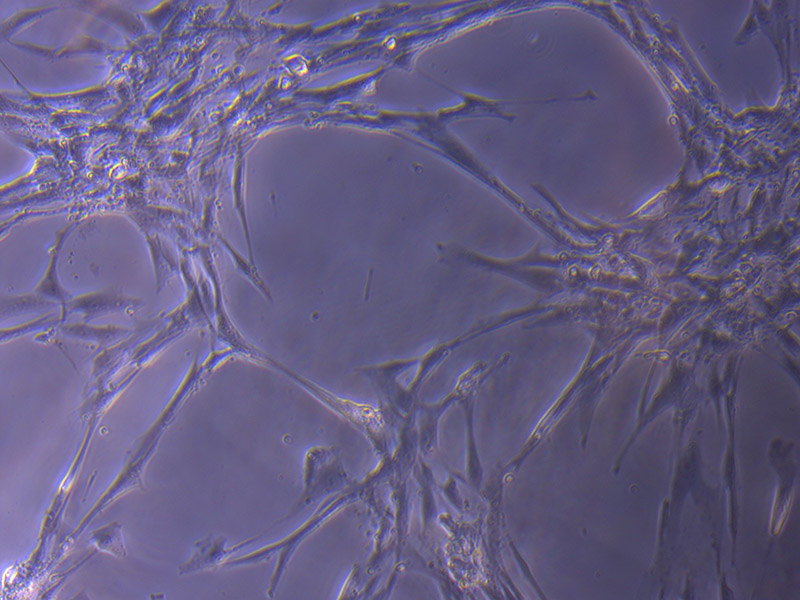


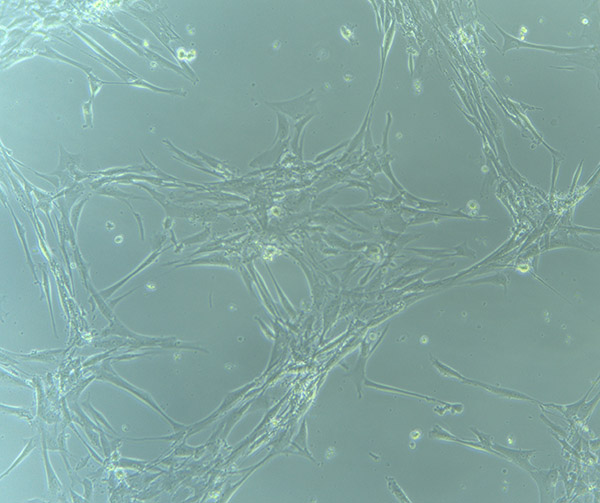
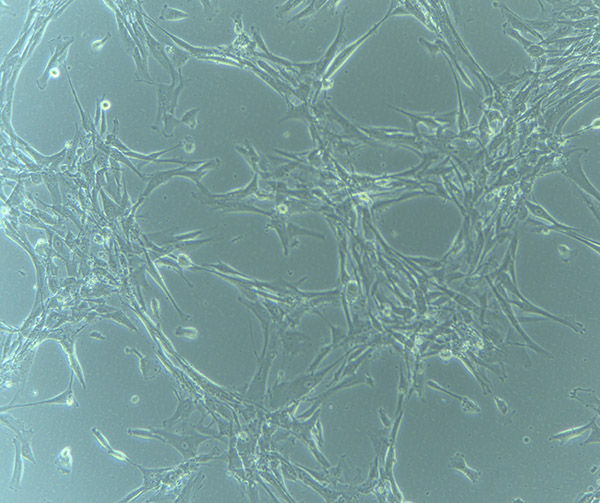 HBVPs growing in T75 Flask – 13/9/21
HBVPs growing in T75 Flask – 13/9/21
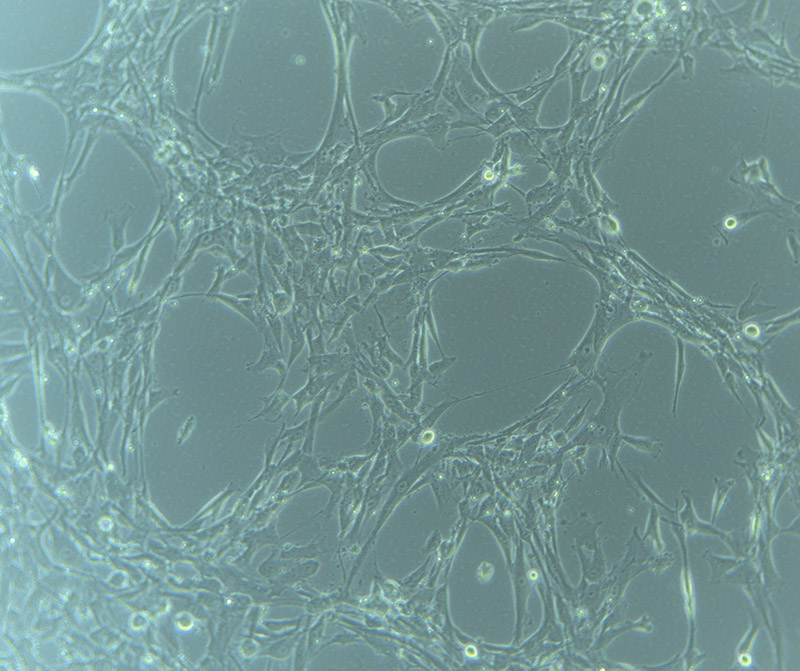

 I engraved the base of two dishes with a ripple pattern to see if the engraving would impact on the growth/adherence of the cells. Inspired by the awesome work of
I engraved the base of two dishes with a ripple pattern to see if the engraving would impact on the growth/adherence of the cells. Inspired by the awesome work of